Metals melt, as a rule, at a very high temperature, which can reach more than 3 thousand degrees. Although some of them can be melted at home, such as lead or tin. But mercury is melted at a temperature of minus 39 degrees. This cannot be achieved at home. Melting point is one of the important indicators of the production of not only the metal itself, but also its alloys. When smelting raw materials, specialists take into account other physical and chemical properties of the ore and metal.
Iron and its properties
Iron is a chemical element that is number 26 on the periodic table. It is one of the most abundant elements in the entire solar system. According to research materials, the Earth's core contains approximately 79−85% of this substance . There is also a large amount of it in the earth's crust, but it is inferior to aluminum.
In its pure form, the metal is white with a slightly silvery tint. It is plastic, but the impurities present in it can determine its physical properties. Reacts to a magnet.
Iron is present in water. In river waters its concentration is approximately 2 mg/l of metal. In sea water its content can be a hundred or even a thousand times lower.
Iron oxide is the main form that is mined and found in nature. Iron oxide can be located in the uppermost part of the earth's crust and be a component of sedimentary formations.
An element in twenty-sixth place on the periodic table can have several oxidation states. It is they who determine its geochemical feature of being in a certain environment. In the Earth's core, the metal is present in a neutral form.
Is metal melting physical or chemical?
Is the melting of graphite and metal a physical or chemical phenomenon? Why?
Answers and explanations 1
Physical phenomena, because melting under fire causes deformation. These are physical phenomena.
Do you know the answer? Share it!
How to write a good answer?
To add a good answer you need:
- Answer reliably those questions to which you know the correct answer;
- Write in detail so that the answer is comprehensive and does not prompt additional questions;
- Write without grammatical, spelling and punctuation errors.
- Copy answers from third-party resources. Unique and personal explanations are well appreciated;
- Answer not to the point: “Think for yourself,” “Easy,” “I don’t know,” and so on;
- Using swear words is disrespectful to users;
- Write in UPPER CASE.
Mining
There are several ores containing iron. However, the following are mainly used as raw materials for iron production in industry:
- magnesite ore;
- goethite ore;
- hematite ore.
And also the following types of ore are often found:
- lellingitis;
- siderite;
- marcasite;
- ilmenite;
- is violent.
There is also a mineral called melanterite . It is used primarily in the pharmaceutical industry. It consists of green, fragile crystals with a glassy sheen. Medicines containing ferum are produced from it.
The main deposit of this metal is South America, namely Brazil.
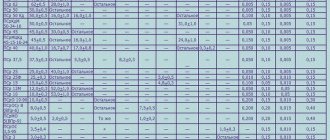
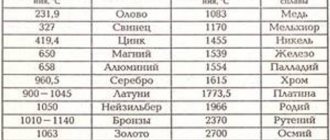
Melting point table
It is important for anyone involved in the metallurgical industry, whether a welder, foundry worker, smelter or jeweler, to know the temperatures at which the materials they work with melt. The table below shows the melting points of the most common substances.
Read also: How to make a screw jack with your own hands
Table of melting temperatures of metals and alloys
| Name | T pl, °C |
| Aluminum | 660,4 |
| Copper | 1084,5 |
| Tin | 231,9 |
| Zinc | 419,5 |
| Tungsten | 3420 |
| Nickel | 1455 |
| Silver | 960 |
| Gold | 1064,4 |
| Platinum | 1768 |
| Titanium | 1668 |
| Duralumin | 650 |
| Carbon steel | 1100−1500 |
| Cast iron | 1110−1400 |
| Iron | 1539 |
| Mercury | -38,9 |
| Cupronickel | 1170 |
| Zirconium | 3530 |
| Silicon | 1414 |
| Nichrome | 1400 |
| Bismuth | 271,4 |
| Germanium | 938,2 |
| Tin | 1300−1500 |
| Bronze | 930−1140 |
| Cobalt | 1494 |
| Potassium | 63 |
| Sodium | 93,8 |
| Brass | 1000 |
| Magnesium | 650 |
| Manganese | 1246 |
| Chromium | 2130 |
| Molybdenum | 2890 |
| Lead | 327,4 |
| Beryllium | 1287 |
| Will win | 3150 |
| Fechral | 1460 |
| Antimony | 630,6 |
| titanium carbide | 3150 |
| zirconium carbide | 3530 |
| Gallium | 29,76 |
In addition to the melting table, there are many other supporting materials. For example, the answer to the question what is the boiling point of iron lies in the table of boiling substances. In addition to boiling, metals have a number of other physical properties, such as strength.
Iron melting and required temperature
The melting point of a metal is the minimum temperature at which it changes from solid to liquid. At the same time, it remains practically unchanged in volume.
Metal can be produced from ore in various ways, but the most basic of them is blast furnace . In addition to blast furnace, iron smelting is also used by roasting crushed ore with an admixture of clay. From the resulting mixture, pellets are formed, which are processed in a furnace followed by reduction with hydrogen. Next, the iron is melted in an electric furnace.
The melting point of iron is very high. For a technically pure element it is +1539 °C. This substance contains an impurity - Sulfur, which can only be extracted in liquid form. Without impurities, pure material is obtained by electrolysis of metal salts.
Metal melting process
This process refers to the transition of a substance from a solid to a liquid state. When the melting point is reached, the metal can be in either a solid or liquid state; further increase will lead to the complete transition of the material into a liquid.
The same thing happens during solidification - when the melting point is reached, the substance will begin to transition from a liquid to a solid state, and the temperature will not change until complete crystallization.
It should be remembered that this rule applies only to pure metal. Alloys do not have a clear temperature boundary and undergo state transitions in a certain range:
- Solidus is the temperature line at which the most fusible component of the alloy begins to melt.
- Liquidus is the final melting point of all components, below which the first alloy crystals begin to appear.
It is impossible to accurately measure the melting point of such substances; the point of transition of states is indicated by a numerical interval.
Depending on the temperature at which metals begin to melt, they are usually divided into:
- Low-melting, up to 600 °C. These include tin, zinc, lead and others.
- Medium melting, up to 1600 °C. Most common alloys, and metals such as gold, silver, copper, iron, aluminum.
- Refractory, over 1600 °C. Titanium, molybdenum, tungsten, chromium.
There is also a boiling point - the point at which the molten metal begins to transition into a gaseous state. This is a very high temperature, typically 2 times the melting point.
Effect of pressure
The melting temperature and the equal solidification temperature depend on pressure, increasing with its increase. This is due to the fact that with increasing pressure the atoms come closer to each other, and in order to destroy the crystal lattice they need to be moved away. At increased pressure, greater thermal energy is required and the corresponding melting temperature increases.
There are exceptions when the temperature required to transform into a liquid state decreases with increased pressure. Such substances include ice, bismuth, germanium and antimony.
Classification of metals by melting point
Different metals can turn liquid at different temperatures. As a result, a certain classification is distinguished. They are divided as follows:
- Low-fusibility elements are those elements that can become liquid even at temperatures below 600 degrees. These include zinc, tin, lead, etc. They can be melted even at home - you just need to heat them up using a stove or soldering iron. Such types have found application in technology and electronics. They are used to connect metal elements and move electric current. Tin melts at 232 degrees, and zinc at 419 degrees.
- Medium-melting - elements that begin to melt at temperatures from six hundred to one thousand six hundred degrees. These elements are used mainly for building elements and metal structures, that is, when creating fittings, slabs and building blocks. This group includes: iron, copper, aluminum. The melting point of aluminum is relatively low and is 660 degrees. But iron begins to turn into a liquid state only at a temperature of 1539 degrees. It is one of the most common metals used in industry, especially in the automotive industry. However, iron is susceptible to corrosion, that is, rust, so it requires special surface treatment. It must be coated with paint or drying oil, and moisture must not be allowed to enter.
- Refractory are materials that melt and become liquid at temperatures above 1600 degrees. This group includes tungsten, titanium, platinum, chromium, etc. They are used in the nuclear industry and for some machine parts. They can be used for melting other metals, making high-voltage wires or wire. Platinum can be melted at 1769 degrees, and tungsten at 3420 °C.
The only element that is in a liquid state under normal conditions is mercury. Its melting point is minus 39 degrees and its vapors are poisonous, so it is used only in laboratories and closed containers.
Read also
Wood structure
Wood structure By making only a cross section, you can clearly see the structure of the wood. Each block of unhewn wood has bark - this is the skin of the tree, which is not used in work; it must be removed. Beneath the bark is the tree's growth zone, which
Wood structure
Wood structure By making only a cross section, you can clearly see the structure of the wood. Each block of unhewn wood has bark - this is the skin of the tree, which is not used in work; it must be removed. Beneath the bark is the tree's growth zone, which
LECTURE No. 1. Structure of wood
LECTURE No. 1. Structure of wood 1. Types of tree species and parts of wood Growing trees have the following components: roots, trunk, branches, leaves. The root system of trees acts as a supplier of moisture and nutrients from the soil along the trunk and branches to the leaves.
Macroscopic structure of wood
2. Macroscopic structure of wood With a cross section of a tree trunk, the main macroscopic features can be established: sapwood, core, annual layers, pith rays, vessels, resin ducts and pith repetitions. Young trees of all species have wood
Structure of metals
1. Structure of metals Metals and their alloys are the main materials in mechanical engineering. They have many valuable properties, mainly due to their internal structure. A soft and ductile metal or alloy can be made hard and brittle, and vice versa. In order to
Properties of glass melts
Properties of glass melts The properties of glass melts include viscosity, the associated hardening rate, surface tension and crystallization, as well as heat capacity, thermal conductivity, and electrical conductivity. The significance of these properties in production
§ 3.3 The structure of atoms and Mendeleev’s periodic law
§ 3.3 The structure of atoms and Mendeleev’s periodic law The properties of simple bodies, as well as the forms and properties of compounds of elements, are periodically dependent (or, expressed algebraically, form a periodic function) on their atomic weights. DI. Mendeleev It is believed that
§ 3.6 Structure of nuclei
§ 3.6 Structure of nuclei The more nucleons must fit in the nucleus, the greater the surface area of the nucleus, where protons and neutrons are added... These features are best met by the shape of the nucleus in the form of two Cheops pyramids connected
§ 4.14 Structure of matter and chemical bonding
§ 4.14 The structure of matter and the chemical bond What finally appears to us as hardened and dense must undoubtedly consist of hooked principles, linked together like intertwined branches. In this category of things, taking first place in it, There will be diamonds
Chapter 32 The Structure of Space – Time
Chapter 32 Structure of Space - Time “Action is the curvature of the World” Pavel Dmitrievich Uspensky, 1911 We have already assumed analogies of the quantum structure of the microworld and the macroworld, under certain conditions. Next, the laws of the resonant structure of our
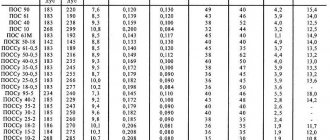
Strength of metals
In addition to the ability to transition from a solid to a liquid state, one of the important properties of a material is its strength - the ability of a solid body to resist destruction and irreversible changes in shape. The main indicator of strength is the resistance that occurs when a pre-annealed workpiece breaks. The concept of strength does not apply to mercury because it is in a liquid state. The designation of strength is accepted in MPa - Mega Pascals.
There are the following strength groups of metals:
- Fragile. Their resistance does not exceed 50MPa. These include tin, lead, soft-alkaline metals
- Durable, 50−500 MPa. Copper, aluminum, iron, titanium. Materials of this group are the basis of many structural alloys.
- High strength, over 500 MPa. For example, molybdenum and tungsten.
Metal strength table
| Metal | Resistance, MPa |
| Copper | 200−250 |
| Silver | 150 |
| Tin | 27 |
| Gold | 120 |
| Lead | 18 |
| Zinc | 120−140 |
| Magnesium | 120−200 |
| Iron | 200−300 |
| Aluminum | 120 |
| Titanium | 580 |
The most common alloys in everyday life
As can be seen from the table, the melting points of elements vary greatly even among materials commonly found in everyday life.
Thus, the minimum melting point of mercury is -38.9 °C, so at room temperature it is already in a liquid state. This explains why household thermometers have a lower mark of -39 degrees Celsius: below this indicator, mercury turns into a solid state.
The most common solders in household use contain a significant percentage of tin, which has a melting point of 231.9 °C, so most solders melt at the operating temperature of the soldering iron 250−400 °C.
In addition, there are low-melting solders with a lower melt limit, up to 30 °C, and are used when overheating of the materials being soldered is dangerous. For these purposes, there are solders with bismuth, and the melting of these materials lies in the range from 29.7 - 120 °C.
Melting of high-carbon materials, depending on alloying components, ranges from 1100 to 1500 °C.
The melting points of metals and their alloys are in a very wide temperature range, from very low temperatures (mercury) to several thousand degrees. Knowledge of these indicators, as well as other physical properties, is very important for people who work in the metallurgical field. For example, knowledge of the temperature at which gold and other metals melt will be useful to jewelers, foundries and smelters.
Metals melt, as a rule, at a very high temperature, which can reach more than 3 thousand degrees. Although some of them can be melted at home, such as lead or tin. But mercury is melted at a temperature of minus 39 degrees. This cannot be achieved at home. Melting point is one of the important indicators of the production of not only the metal itself, but also its alloys. When smelting raw materials, specialists take into account other physical and chemical properties of the ore and metal.
Read also: DIY 12 volt switching power supply
Features of melting non-ferrous metals and alloys
Interaction of metallurgical melts with gases and lining materials of melting furnaces
In most cases, non-ferrous metals are melted in air. Metal melts interact with the atmosphere and refractory materials; during melting, they can become contaminated with various types of impurities.
The gaseous medium with which the metal melt comes into contact during melting and casting is not only ordinary air, but also the combustion products of organic fuel, various specially used gases (argon, nitrogen, helium), i.e. the gas environment above the melt may contain oxygen, nitrogen, water vapor, carbon oxides (CO and CO2), sulfur dioxide SO2, methane CH4.
The interaction of a metal melt with a gaseous medium can be of three types:
- first type - the melt does not interact with gases and does not dissolve gas in itself (complete inertness); This is how all liquid metals behave in an atmosphere of inert gases (argon, helium, etc.). Almost complete inertness is observed between many elements and nitrogen (copper - nitrogen, zinc - nitrogen, etc.), as well as hydrogen (tin - hydrogen, lead - hydrogen) and oxygen (gold - oxygen);
- second type - a stable metal-gas compound is formed; this occurs between oxygen and liquid aluminum, magnesium, zinc, tin. The melt becomes contaminated with particles of these compounds;
- third type - gas solutions are formed in the melt; A similar type of interaction is observed in many systems metal - hydrogen (Mg, Al, Cu, Ni, Fe), metal - oxygen (Cu, Ni, Fe).
The interaction of basic non-ferrous metals with gases is presented in Table. 1.
Table 1
Formation of solutions of gases and gas-forming elements in liquid metals
Gas or gas-forming element Metal
| Mg | Al | Ag | Cu | Mn | Ni | Fe | Ti | Cr | |
| Hydrogen | + | + | + | + | + | + | + | + | + |
| Oxygen | — | — | + | + | + | + | + | + | + |
| Nitrogen | — | — | — | — | + | + | + | + | + |
| Carbon | — | — | — | — | + | + | + | + | + |
| Sulfur | — | + | + | + | + | + | + | + | + |
| Note: The sign (-) indicates the lack of solubility of gases; sign (+) indicates practically significant solubility | |||||||||
Hydrogen usually constitutes the major proportion of gases found dissolved in metals. The main source of hydrogen is water, which reacts with the melt according to the reaction Me + H2O - MeO + 2[H]Me, i.e. the metal reduces hydrogen, which in the atomic state easily dissolves in the melt (Table 1). Water is contained in the charge, lining, and slag.
Oxygen actively interacts with most metals, forming oxides and oxygen solutions. If oxygen is in dissolved form, then deoxidation is necessary, i.e. converting oxygen into insoluble form. Insoluble oxides are removed from the melt by settling, filtering, etc.
Low-melting metals , including Mg and Al, do not dissolve oxygen (Table 1); their interaction is expressed only in the formation of a film of insoluble oxides on the surface of the melts. Alloys based on low-melting metals with any alloying elements behave with oxygen like pure metals.
A peculiarity of the behavior of oxygen solutions in liquid metals during melting in air is that during cooling and crystallization of the melt, this gas is never released in free form, but passes either entirely into a solid solution (Ti-O system) or into solid compounds - oxides (Cu-O, Fe-O systems).
Nitrogen does not dissolve in fusible metals (Table 1). More refractory metals, starting with manganese, dissolve nitrogen, and in Mn, Ni and Fe, when the temperature decreases, especially during crystallization, the solubility of nitrogen in metals decreases, and this can cause the appearance of gas porosity of nitrogen origin. In melts of Ti, Vi and Cr saturated with nitrogen, on the contrary, as the temperature decreases, nitrogen completely passes into solid solution (Ti-N) or forms nitrides (Cr-Ni).
Nitrogen remaining in the solid metal causes a sharp decrease in the ductility of the metal, especially if it forms brittle nitride phases.
Water vapor interacts with almost all metals at temperatures when they are in a liquid state. Low-melting metals do not dissolve either oxygen or hydrogen. Consequently, in the liquid state, these metals will be oxidized by water vapor and covered with a film of insoluble oxides. Liquid Mg and Al will dissolve the hydrogen released. Therefore, melting these metals in an atmosphere containing harmful vapors will cause not only contamination of the melt with non-metallic inclusions of an oxide nature, but also saturate it with hydrogen. All other metals (Table 7) are capable of dissolving both oxygen and hydrogen. Therefore, when melting in an atmosphere containing water vapor, they will be contaminated with both hydrogen and oxygen.
Carbon monoxide CO interacts with low-melting metals (up to aluminum) according to the reaction Me + CO = MeO + C. More active metals from this group (Mg, Al) will oxidize in a CO atmosphere at normal melting temperatures. Therefore, the atmosphere of this gas for a given metal is oxidizing, causing contamination of the melt with non-metallic inclusions.
Liquid copper and silver are capable of dissolving oxygen, but the melts are stable in contact with CO. This is explained by the fact that the reaction between the metal and CO practically does not occur. Carbon monoxide is a reliable protective medium when melting these metals.
All other liquid metals listed in table. 1, both oxygen and carbon dissolve. Therefore, during melting, they are saturated with dissolved carbon and oxygen, and during crystallization, porosity formed by carbon oxide appears in the metal.
Carbon dioxide (CO2) practically does not interact with liquid metal, which has a low affinity for oxygen ( Bi,
Pb , Ag, C u
). Therefore, these elements can be melted in a CO2 environment. For all other metals, pure CO2 is an oxidizing gas.
Sulfur dioxide (SO)2 with metals that do not dissolve oxygen, but dissolve sulfur (Sn, Ps, Al), interact according to the reaction
2Me + SO2 ↔ 2MeO+ [S]
Consequently, the melts of these metals will be contaminated with oxide inclusions and dissolved sulfur. For metals capable of dissolving oxygen and sulfur (Ag, Cu, Mn, Ni, Fe), the reaction with SO2 can be written as follows:
Me + SO2 ↔ Me+ [S] + 2[O]
As the temperature decreases for all metals, the equilibrium shifts to the right, towards the decomposition of SO2. The exceptions are copper and silver, for which a decrease in temperature shifts the equilibrium to the left, towards the formation of SO2.
Methane (CH4) may be present in the atmosphere of gas furnaces that burn fuel oil and natural gas. At temperatures above 1000°C, methane dissociates into elements, which can cause the melt to become saturated with hydrogen and carbon simultaneously.
Liquid metals, while in melting furnaces and crucibles, come into contact with the materials used for lining the baths of melting furnaces and pouring ladles, as well as for the manufacture of melting crucibles. These materials can be divided into oxide, oxide-graphite, pure graphite, carborundum, and metal.
The most dangerous is the interaction of the metal melt with the lining oxide according to the reaction Me + RO↔ Me+ [R]. With this interaction, not only the lining is destroyed, but the melt is also contaminated with the reduced element R and Me oxides or dissolved oxygen.
Oxide-graphite materials, which are a mixture of fireclay with 30 - 40% graphite, have a fire resistance of 1300 - 1400 oC and are used for melting metals that do not interact with C and SiO2. Fireclay-graphite crucibles are used for melting aluminum, copper and their alloys, as well as alloys based on gold and silver.
Graphite burns out very quickly in air, starting from 600 - 700°C, although by itself it is capable of working up to 2500°C. Graphite refractories are suitable for melting low-melting metals. Most often, graphite is used for melting copper and its alloys, however, starting from 600°C, protection against their oxidation is necessary.
Carborundum materials (~ 80% SiC, the rest are refractory oxides) are very fire-resistant (~ 2000°C) and chemically resistant. Carborundum lining is used for melting aluminum and copper alloys, which practically do not react with SiC.
Metal materials are used for the manufacture of melting tanks intended for smelting relatively low-melting materials. These materials are mainly cast iron and steel. The main type of interaction of melts with such materials is simple dissolution of iron, and the contamination of some metals with iron can be quite large, for example: at 700 °C 2.5% Fe can dissolve in aluminum. To prevent saturation of the melts with iron, the inner surface of the crucibles is painted with a special composition.
Thus, the melts obtained during melting may contain various impurities that are introduced by the starting materials (charge) and appear during melting as a result of interaction with the atmosphere and refractory materials.
Impurities in metal melts can be present in dissolved form and in the form of insoluble suspended particles. Soluble impurities include:
-metals (Pb, Sn, Al in alloys based on iron, nickel and copper; Fe in alloys based on aluminum and magnesium);
-non-metals (S, P, Si, C, etc.);
-gases (H, O, N).
Insoluble impurities include:
-non-metallic inclusions (oxides, nitrides, etc.);
- inclusions of metallic nature (intermetallic compounds).
Protection of melts from interaction with the atmosphere
If, during interaction with air, insoluble compounds are formed on the surface of the melt and the resulting film of these compounds slows down further interaction, then in this case the melting is carried out in direct contact with the atmosphere (for example, the melting of most aluminum alloys). If the resulting film is fragile and unable to protect the melt from further interaction, then special measures are taken using fluxes, slags or protective atmospheres.
Protective fluxes and slags are complex alloys of salts and oxides loaded onto the surface of the melt. Fluxes are introduced to form liquid slags that cleanse the metal of unwanted impurities. Depending on their purpose, fluxes are divided into covering (protective), refining and universal. Fluxes and slags must be more fusible than the metal melt being protected, less dense than liquid metal and impermeable to air gases. For example, protective slag for melting copper alloys consists of 73% SiO2 and 27% NaO and melts at 795 ° C (an aqueous solution of this compound is called liquid glass). The disadvantage is evaporation.
In cases where it is impossible or undesirable to use slag or flux, for example, when preparing complex alloys with chemically active additives, and also because of the danger of corrosion of the lining and contamination of the melt, protective atmospheres, such as argon, are used, or vacuum melting is used (this is the most reliable method of obtaining pure metals). But, despite the protection, metal melts are still contaminated with various impurities of metals and non-metals, soluble and insoluble in the melt.
Refining (purification) of melts
Soluble impurities are removed by chemical methods, insoluble (non-metallic inclusions) impurities are removed by mechanical methods.
Refining of melts from dissolved impurities is carried out during the preparation of the alloy by oxidation, chlorination, treatment with fluxes or vacuum distillation.
Refining by oxidation is used in cases where the alloy is capable of dissolving oxygen. This method can clean the melt from impurities that have a greater affinity for oxygen than the main component of the alloy being refined.
Oxidative refining is carried out by blowing with air or introducing oxidizing agents into the melt. When the melt comes into contact with oxygen, first of all, oxidation of the base metal occurs, and the melt is saturated with oxygen. After this, dissolved impurities combine with oxygen to form insoluble oxides, which turn into slag.
Chlorination is the purging of melts with chlorine gas. It is used to remove impurities that have a greater affinity for chlorine than the base metal and the main alloying components of alloys (for example, removing Na and Mg impurities from aluminum alloys).
Refining by fluxing is used to remove impurities that form volatile or easily slagging compounds with flux that are not soluble in the base metal (for example, cleaning aluminum alloys from magnesium with cryolite).
Refining by vacuum distillation is used to remove impurities that have a higher vapor pressure than the base metal (for example, removing zinc from aluminum).
Insoluble impurities can have a wide variety of sizes - from fractions of a micrometer to millimeters. These are mainly oxide compounds, although there may be nitrides, carbides, and carbonitrides. They are called non-metallic inclusions. They sharply reduce the ductility of metals, especially under shock loads and low temperatures, and also significantly reduce the corrosion resistance of alloys.
The sources of these inclusions are particles of the lining, slag, flux, charge, and products of physical and chemical processes occurring in the melt.
Non-metallic inclusions can be removed by settling, blowing with gases, vacuuming, treatment with fluxes and slags, and filtration.
Settling is usually a lengthy process. Large particles (more than 100 microns) are separated in a few minutes, medium ones (10 - 100 microns) - in tens of minutes, and small ones (less than 5 microns) are almost impossible to separate by settling. This technique is not capable of deep cleaning, but due to its accessibility it is often used.
The essence of refining by blowing with gases or treating with volatile substances is that gas bubbles, passing through the melt, capture foreign inclusions encountered along the way and bring them to the surface of the melt. Gas purging allows you to remove both large and small (up to 1 micron) particles. Argon or nitrogen is usually used for purging. Sometimes chlorine is added to argon (for example, when blowing aluminum alloys). Chlorine destroys the oxide film on the inner surface of the gas bubble, resulting in improved adhesion of the bubbles to the particles. Instead of gases for refining aluminum alloys, volatile solid compounds are also used - aluminum and manganese chlorides, zinc chloride, hexachloroethane, etc. When processing melts, aluminum chloride AlCl3 and ammonium chloride NH4Cl are sublimated, since they have a low sublimation temperature: the first is 180 ° C, the second is 338 ° C. Manganese and zinc chlorides interact with the melt:
3MnCl2 + 2Al— 2 AlCl3 + 3Mn, 3ZnCl2 + 2Al— 2AlCl3 + 3Zn.
In practice, vapors of aluminum chlorides are purged.
Vacuum refining uses the floating action of gas bubbles released from the solution. The residual pressure during evacuation is 500 - 1000 Pa.
Refining by flux treatment is based on the transition of particles of undissolved impurities into slag or flux as a result of dissolution or wetting. Fluxes contain increased amounts of fluoride salts and sodium oxide in the form of Na2CO3. This method requires active mixing of the melt with flux or slag. After refining, settling is carried out to allow drops of slag or flux to float to the surface.
The most effective way to remove particles of insoluble impurities from the melt is filtration.
Refining melts from dissolved gases (degassing) is the removal of hydrogen, nitrogen, and carbon monoxide from melts. Removal of dissolved oxygen is carried out by deoxidation. Degassing is carried out by freezing, blowing with gases, vacuuming, flux treatment, and various physical effects on the metal.
Freeze degassing is based on a decrease in the solubility of gases with decreasing temperature. In the furnace, the melt is slowly cooled (up to partial crystallization), while dissolved gases are released into the atmosphere through the open surface of the melt. After this, the metal is heated again at maximum speed. This method is very time-consuming, but quite effective.
Degassing of melts by blowing with insoluble gases is based on the fact that in a bubble of such gas the partial pressure of the gas dissolved in the melt is zero and the dissolved gas passes from the melt into the bubble. In this case, a large bubble surface is required, i.e. their smallest size is desirable (no more than 0.1 - 0.5 mm).
Argon (all alloys), nitrogen (copper and aluminum alloys), nitrogen with chlorine (aluminum alloys) are used for purging. Sometimes volatile compounds are used - manganese chloride (for copper and aluminum alloys), zinc chloride and hexachloroethane (for aluminum alloys).
The most reliable way to degas metal melts is by vacuuming. A decrease in pressure above the melt leads to the release of gases not only through the surface of the melt, but throughout the entire volume in the form of bubbles. The centers for the release of dissolved gas are tiny vapors and cracks on the surface of impurity particles that are always present in melts. Therefore, along with gas bubbles, impurity particles float to the surface of the melt.
When processing melts with slags and fluxes, degassing also occurs. However, the mechanism of this effect is not fully understood.
It has been established that some physical influences on the melt (for example, ultrasonic vibrations or direct current treatment) lead to degassing. The exact nature of these processes has not yet been clarified.
Deoxidation of metal melts
Deoxidation (removal of dissolved oxygen) is carried out only when melting those metals and alloys that are capable of dissolving oxygen while in a liquid state. Of the non-ferrous metals, nickel and nickel alloys with copper, iron, chromium, and manganese are subjected to mandatory deoxidation during conventional smelting in air. Pure copper and copper alloys with silver and nickel also need to be deoxidized. Copper alloys with oxygen-active metals such as Zn, Al, Cr, Zr are almost incapable of dissolving oxygen and deoxidation makes no sense for them.
Alloys based on aluminum, magnesium, zinc, tin, and lead never deoxidize. Oxygen is present in the melts of these metals and their alloys in a bound state in the form of insoluble non-metallic inclusions.
Modification
Modification is a change in the structure of a solid metal due to the introduction of special additives or as a result of certain conditions of melting and processing of the melt. During modification, as a rule, the structure is refined. This leads to improved technological ductility and increased mechanical properties.
Modification can be done by overheating the melt and rapidly cooling it. This modification is very effective for many aluminum and copper alloys. The application of mechanical or ultrasonic vibrations to the crystallizing melt also leads to refinement of the structure. The so-called suspension casting has been developed - this is a type of modification by dissolving a molten hard alloy of the same composition in a stream.
The most common method of modification is the introduction of additives - modifiers. These additives either act as additional crystallization centers (for example, AlP, Al3Ti, AsSc, etc.) or impede crystal growth (for example, Na in aluminum alloys).
Modification using additives is carried out at the end of the melt, since the modifying effect of additives is limited in time. The amount of additives does not exceed 0.1% by weight of the melt.
Source: L.P. Kochetkova “Metallurgical processes in mechanical engineering” Kirov 2004