Various anti-corrosion protection technologies are used for metal sheets and parts. Cathodic protection against corrosion has become widespread. This method has a number of characteristic features, and most often cathodic protection is used for large objects. These could be pipes, cars, metal pile structures, sea vessels. How exactly are pipelines protected from corrosion at the physical and chemical level?
Basic cathodic protection technologies
Cathodic protection is a special method of electrochemical protection of metal objects from rust and corrosion. The main principle is that a negative electrical potential is applied to the metal object being protected. This minimizes metal contact with external ions and substances with an electrical charge. The technology was developed approximately 200 years ago by British scientist Humphry Davy. To confirm his theory, he compiled several reports that were submitted to the government. Based on these reports, the world's first cathodic protection of a large industrial ship was carried out.
Anti-corrosion protection applies to various objects - pipelines, cars, roads, airplanes and so on. Please note that the type of metal does not matter - it can be iron, copper, silver, gold, aluminum, titanium and any other metal, as well as various alloys (with or without alloying additives). Corrosion protection of a car, individual pipe fragments, various decorative items of complex shape, and so on can be equally successful.
1 way
Connecting the part to an external source of electric current (usually compact substations perform this role). When the technology is used, the metal object acts as a cathode, and the electrical substation acts as an anode. Due to this, a shift in the electrical potential occurs, which makes it possible to protect the metal object from electrically active particles. The main areas of application of this technology are the protection of pipelines, welded structures, various platforms, road surface elements, and so on. This technology is quite simple and universal, so it is very popular in the world. Its main disadvantage is the need to connect the protective circuit to an external current source, which can be inconvenient in the case of objects that are located far from human civilization (this problem is partly solved through the use of autonomous energy sources).
Method 2
Galvanic polarization method (galvanic anode technology). This technique is also quite simple and intuitive: a metal object is attached to another that has a negative charge (most often this element is made of light metals - aluminum, zinc, magnesium). Galvanic polarization technology is usually used in cases where there is a protective layer on the surface of the object. This technology is popular in America, where there are a large number of sparsely populated areas and where there is a shortage of external energy sources. Experts argue that galvanic polarization could become very popular in Russia due to the peculiarities of our geography if a protective coating were applied to domestic pipelines (in such a scenario, the use of the first technology would be very difficult, forcing people to look for an alternative).
Fighting metal corrosion
The choice of the optimal method of protecting metal structures, as well as products made from a particular type of metal, must be made in accordance with a number of certain factors.
These factors include:
- operational features of metal or metal structure;
- characteristics of the metal itself or metal structure;
- climatic conditions of the corresponding region and the like.
Below are the main directions of methods for protecting metals from corrosion processes, which are widely used in industry, production, and also in everyday life:
- structural method;
- passive method;
- active method.
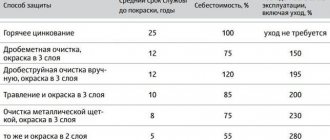
In the first case, to prevent corrosion, non-ferrous metals, stainless steels, and Corten steels are chosen for structural materials. And with the help of special adhesives, sealants and rubber gaskets, designers try to provide maximum protection against the entry of corrosive environments into metal structures.
A passive method of protecting metals from corrosion involves applying a certain coating (enamel, paint, varnish, etc.) in order to prevent the onset of the corrosion process. This method is quite accessible to a wide range of people.
But here you need to understand that various coatings only provide a barrier to the corrosion process, but do not exclude its occurrence. Therefore, it is very important with this method of protecting metals from corrosion to carefully prepare and treat the surface for painting, apply this or that coating as evenly as possible, maintain a certain layer thickness, strength and ensure the absence of air cavities.
If we talk about coatings, the most common today are: paint without rust removal and liquid plastic.
The use of anti-rust paint is the most popular method of protecting metal, as it performs a number of basic functions: transforming rust, combining an anti-corrosion primer and top enamel.
Enamel is very popular due to its wear resistance and weather protection. The paint can be applied to both clean and corroded surfaces.
The method of protecting metals using liquid plastic is relatively new, effective and very simple. It is used for painting pipelines, gratings, metal furniture and other structures. Liquid plastic can be applied to any uncleaned surface at any level of corrosion, and you don’t even have to pre-clean automotive parts or any other structures.
Silver is a noble precious metal, but silver products have a big drawback - they quickly become covered with plaque and blackness. Find out all the secrets of cleaning silver at home and never face this problem.
Even the ironing surface of non-stick irons can develop carbon deposits over time. Here we describe how to clean a steam iron without damaging the Teflon coating.
Is your enamel kettle covered with a thick layer of limescale? Using simple household cleaning products, you can easily descale an enamel kettle, read more here.
The main advantage of this method is the ability to use any synthetic wet cleaning products.
Cathode polarization technology
In this case, the so-called superimposed current is used. An external conductor (often) or a current source (rarely) is used to apply it to a metal object. Upon contact with an electrically active particle, the following happens: the particle, under the influence of electrical attraction forces, moves to a protective element with a negative charge, where “recycling” of these particles occurs.
The consequences of such “disposal” are obvious - the protective element itself becomes corroded over time and becomes unusable. Therefore, this technology is often called the sacrificial electrode method (instead of our part, the “sacrificial electrode” rusts).
In addition to current and voltage, when working with cathodic polarization, one more important parameter must be taken into account - the ohmic voltage. In a technical sense, this parameter reflects the fact that as an electrical charge flows over time, the current voltage in the circuit drops. The drop itself occurs due to the fact that the cathode current flows along a circuit with a lower charge. If the circuit is assembled correctly, this indicator is quite small - thanks to this, the same current of the same power will always be maintained in the circuit.
Stress Corrosion Cracking
If a metal surface is simultaneously exposed to external negative factors and high voltage from power lines, which creates tensile forces, then rust formation occurs. According to the research carried out, the new hydrogen-corrosion theory gained its place.
Small cracks are formed when the pipe is saturated with hydrogen, which then ensures an increase in pressure from the inside to levels higher than the required equivalent of the bond of atoms and crystals.
After the crack opens, the rusting process of the metal accelerates, which is provided by the ground electrolyte. As a result, under the influence of mechanical influences, the metal undergoes slow destruction.
Technology for creating protection stations
Another technology for creating cathodic protection is connecting the element to external current sources. In most cases, for these purposes, special cathodic protection stations (CPS) are built, which consist of several elements - the main current source, anode grounding, various cables and wires connecting individual structural elements and auxiliary points with mechanical or computer control, which allow you to control the parameters .
Most often, this technology is used for objects located near power lines - these can be pipelines, various factory buildings, and so on. VCS can operate in multi-threaded mode - in this case they will serve several protection systems at once. On pipes, a practice has become widespread in which several separate blocks are placed on the pipes to distribute the current more efficiently. The thing is that in the case of long pipelines, in the places where the pipes are connected to current sources, special points with an increased level of electric field voltage are formed - because of this, damage to the pipes can occur. The use of such blocks allows electricity to be distributed evenly throughout the entire protective circuit.
Automation
Checkpoints can operate both manually and automatically:
- In the case of manual control, the change in voltage parameters is regulated by the operator. At the physical level, regulation is carried out by switching the operation of the transformer. The operation of the winding is regulated, which allows you to change the parameters of the electric current.
- In the case of automatic control, the change in voltage parameters is regulated by the device itself based on the parameters that were once set by the operator. At the physical level, control is carried out using special semiconductor thyristors. They turn on or off when the electric current parameters deviate from the specified parameters.
Features of cathodic protection of pipes
Corrosion in pipelines usually occurs due to various defects and damage to the pipes - ruptures, cracking, cracks, and so on. Due to corrosion, the sealing of pipes is compromised, which can lead to complete or partial failure of the pipeline. This problem is especially acute for underground pipelines. When pipes are placed underground, areas with different electrical potentials are created. This is due to the heterogeneity of the soil and the presence of various debris of inorganic origin in the soil. If there is a serious potential difference, negatively charged ions in the ground begin to react with the metal. This leads to corrosion, which quickly destroys the pipeline.
Electric potential
Cathodic protection of pipelines against corrosion is carried out according to two standard schemes. Using cathodic polarization and by creating external stations. Pipeline protection should be aimed primarily at reducing the rate of destruction of the pipe material. This is done by reducing the electrical potential of the pipe in comparison with the electrical potential of the soil:
- The electrical potential of most modern pipes is approximately 0.8-0.9 volts.
- It has been experimentally shown that the main rocks of the soil have a potential of approximately 0.5-0.6 volts.
To equalize the electrical potentials, it is necessary to reduce the potential of the pipes by only 0.3-0.4 volts. This allows you to almost completely stop the appearance of rust. If the work is carried out correctly, the rate of natural rusting will be less than 1 mm per year.
Choosing a method
The technology for creating external protection stations is suitable for pipes. In this case, overhead power lines with voltages from 500 to 10,000 volts are used as power sources. The higher the voltage, the more pipes can be serviced. Sometimes there are no such lines in a particular area. In this case, it makes sense to install various generators.
External station technology has one major drawback. To create protection, labor-intensive and complex work will have to be carried out. This significantly increases the cost of creating a pipeline. When working with high voltage, excess electrical voltage may be created at the point of electricity supply - this can cause hydrogen cracking of pipes, so when carrying out installation work, electrical wiring must be done carefully.
Instead of the technology of protective stations, you can use the technique of using galvanic anodes to create a polarization effect. This technology is suitable for soils with low resistivity (up to 50 Ohms per 1 sq. m). If the soil resistivity is very high, then the technology of using galvanic anodes is practically useless due to its low efficiency.
Types of corrosion
Corrosion has a wide distribution and a variety of conditions and environments in which it can begin. Therefore, there is no specific classification of different cases of corrosion yet.
The table shows the extensive classification of corrosion that exists today.
| Condition/environment conducive to the corrosion process | Types of corrosion | |
| By type of aggressive environment | gas corrosion | |
| corrosion in non-electrolytes | ||
| corrosion in electrolytes | ||
| atmospheric corrosion | ||
| underground corrosion | ||
| corrosion due to stray currents | ||
| biocorrosion | ||
| According to the conditions of the corrosion process | contact corrosion | |
| crevice corrosion | ||
| Full immersion corrosion | ||
| partial immersion corrosion | ||
| corrosion during alternating immersion | ||
| intercrystalline corrosion | ||
| friction corrosion | ||
| stress corrosion | ||
| By nature of destruction | complete corrosion | uniform corrosion |
| uneven corrosion | ||
| selective corrosion | ||
| local corrosion | pitting corrosion | |
| pitting corrosion | ||
| corrosion spots | ||
| through corrosion | ||
| intergranular corrosion | ||
| According to the mechanism of the corrosion process | chemical | |
| electrochemical | ||
The huge number of types of corrosion has led to the emergence of an equally large number of methods and techniques to combat each of them. But this issue is not closed and work continues to create new methods that will be more effective.
Features of cathodic protection of cars
Corrosion on cars often appears suddenly. The speed of its spread is very high, since the car has a large number of moving parts. During operation, various small cracks and dents may form in such elements. This significantly increases the risk of corrosion. Cathodic protection of a car against corrosion is usually carried out by redistributing the electrical potential.
Usually special electronic modules are used, which are compact in size and mounted inside the car. Installation of such blocks takes no more than 20 minutes.
Additional processing
It is also worth noting that the cathodic protection method is usually combined with other techniques:
- All main parts of the car are coated with special paints and mastics. They create a protective layer on the metal surface. This layer is electrically neutral. Therefore, upon contact with electrically active substances or ions, rusting does not occur.
- Some vehicle components may be coated with protective cathode plates, which also minimize the risk of rust. Plates are usually used to cover the moving parts that are most likely to crack and become damaged. This is the bottom of the car, rear wheel arches, headlights, interior door surfaces, and so on.