Menu:
- Clad pipes are
- Application
- Welding
- About manufacturing technologies
- About large diameter pipes
- Video
Due to many positive qualities, clad pipes are used in various spheres of life. The uniqueness of the production technology lies in the multi-layer structure of the products.
Cladding Processes
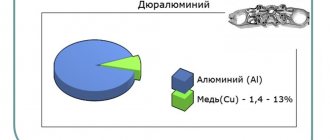
It is carried out in the process of hot rolling (for example, cladding of sheets and plates), pressing (cladding of pipes), as well as by explosion welding. [1] Consists of joint hot rolling or drawing of base and protective metals. The adhesion between metals occurs as a result of diffusion under the influence of joint deformation of the hot workpiece. The protected metal (steel, titanium alloys) is coated on one or both sides with copper, tombac, corrosion-resistant steel, aluminum.
Clad wire is made by drawing a pipe, inside which a core of another metal is inserted.
The purpose of cladding is to create on the surface of a part a layer of material with special properties - high hardness, corrosion and/or wear resistance, etc., it is used in the manufacture of parts/equipment or when restoring their shape after wear. In this case, the thickness of the cladding layer can range from tenths of a millimeter to several millimeters.
The disadvantages of cladding are the high cost of the method and accelerated corrosion in the weld area.
Application
Cladding is used in the manufacture and repair of parts exposed to aggressive environments (dirt, slag, steam) in order to save expensive materials. It is used to produce bimetal and trimetal, to create an anti-corrosion layer of aluminum on sheets, plates, pipes made of aluminum alloys, to apply brass coating to steel sheets (instead of electrolytic coating), etc. Also used in jewelry, for example, gold overlay superimposed on silver (silver with gold).
Reviews
Anna Ivanova: I'm delighted, it just shines. I decided to plate an old ring, my mother’s, with rhodium in order to wear it longer. I paid 800 rubles for a thin ring, but in my opinion, it’s worth it.
Tatyana: And I love such jewelry, it’s especially nice that they shine like gold and cost almost like silver.
Ivan: We applied this coating to wedding rings and noticed that as it wears out, the yellow color of the gold appears. Looks like an amateur. But there really are no noticeable scratches.
Notes
Wikimedia Foundation. 2010.
See what “cladding” is in other dictionaries:
cladding - I, cf. plaquer. specialist. Action by value Ch. clad. Cladding protects metals from corrosion. BAS 1. Cladding, combining silver and gold with copper in the form of thin sheets, for use on various products. Spassky Horn. sl. 1841. That ... Historical dictionary of Gallicisms of the Russian language
PLATING - (plating) (from the French plaquer to apply to cover), applying a thin layer of another metal or alloy to the surface of metal sheets, plates, pipes, wire by hot rolling or pressing (for example, a brass coating on steel ... ... Big Encyclopedic Dictionary
CLADING - CLADING, cladding, many. no, cf. (specialist.). Same as plating. Ushakov's explanatory dictionary. D.N. Ushakov. 1935 1940 ... Ushakov's Explanatory Dictionary
CLADING - covering a sheet of metal during the rolling process with a thin layer of some other metal that is more resistant to corrosion to protect it from destruction. See Alclad. Samoilov K.I. Marine dictionary. M.L.: State Naval... ... Naval Dictionary
cladding - noun, number of synonyms: 2 • application (18) • plating (2) ASIS Dictionary of Synonyms. V.N. Trishin. 2013 ... Dictionary of synonyms
cladding - 1. Applying a thin layer of another metal or alloy to the surface of metal products to protect them from corrosion by a thermomechanical method 2. During excavation work, laying turf on the slopes of embankments and excavations to strengthen them [Terminological... ... Technical Translator's Reference
Cladding is the application of a thin layer of another metal or alloy to the surface of metal products (sheets, plates, wire, pipes, etc.) by a thermomechanical method; hot rolling (for example, cladding of sheets and plates), pressing ... ... Encyclopedic Dictionary of Metallurgy
cladding - plating (from the French plaquer to apply, cover), applying a thin layer of another metal or alloy to the surface of metal sheets, plates, pipes, wire by hot rolling or pressing (for example, brass coating on ... ... Encyclopedic Dictionary
The essence of cladding
Cladding is the application of sheets of another metal to a base metal matrix, as well as hot rolling, which allows the metals to be bonded. During cladding, metal that is unstable to the corrosion process is covered with two sheets of another metal on both sides. Next, the rolling operation is performed.
What does cladding look like in cross section?
Solution
Rhodium plating on silver jewelry is a fairly common way to prevent premature wear. This procedure is often used by jewelers around the world not only to increase the wear resistance of jewelry, but also to give the product a platinum shine and a noble appearance.
The procedure itself is nothing more than sputtering a thin (0.1-25 microns thick) layer of rhodium onto the silver product itself. The cost of this process is already included in the final purchase price. Rhodium is a rare metal whose price is much higher than the price of platinum. It has a higher level of resistance to corrosion and mechanical wear than other precious metals. Rhodium belongs to the group of noble metals due to its spectacular appearance and high chemical resistance. It retains a cold shine for a long time, which cannot be said about silver, and due to its high hardness it wears out much more slowly. Rhodium is a very hard gray metal with a noble silvery tint, which also has a very high level of reflectivity. It is precisely because of these qualities that this metal has not escaped the attention of jewelry manufacturers.
Rhodium plating on silver gives jewelry additional strength and shine, and also greatly prolongs its beauty.
Metals for cladding
- copper alloys;
- carbon steel;
- acid resistant steel.
Metals used as a protective layer:
- stainless steel;
- nickel;
- copper;
- silver;
- gold;
- titanium;
- molybdenum;
- tantalum and others.
The thickness of the cladding layer ranges from 3 to 40% of the thickness of the metal requiring corrosion protection. Clad steel can be easily machined, making it a valuable raw material in industrial production.
The main advantage of cladding technology is that it makes it possible to increase the wear resistance of spare parts and mechanisms. Also, this method of metal processing ensures the rational use of expensive metals and their alloys.
Source of the article: https://dedpodaril.com/lityo/plakirovanie-technoligiy.html
No. 3. How long does gold plating last on silver?
As you already know, the average lifespan of gold plating is two years of constant wear. This means that wedding rings, crosses and chains will have to be updated. In this case, silver will gradually appear through the gold layer - first at the joints, edges of jewelry or places of constant contact with the edge of clothing. It is the uneven color fade that irritates jewelry owners.
The most durable gilding is a layer of 3 microns of 999-carat gold applied to 925-carat silver. Gold adheres securely to silver, is resistant to household chemicals and friction, it is difficult to scratch, does not crumble or peel.
How to wear gold plated? How long gilding on silver lasts depends on how it is handled. To extend the life of the gold plating, remove jewelry when playing sports, before swimming pools and saunas, when cleaning, and when sleeping.
Cleaning gold plated jewelry. It is not difficult to damage the gold layer on silver - for example, with a rough brush or abrasive. Wipe off small stains with an alcohol solution or vinegar on a cotton swab, and large stains with a toothbrush and paste or saline solution.
What is cladding
Not many people who begin to engage in metallurgy know the features of cladding. This is a technological process that involves applying a protective metal coating to the base. For this purpose, the hot rolling method is used. With its help, you can not only apply an additional protective coating, but also strengthen the upper layers of the main part. Most often, steels that are susceptible to corrosion are subjected to cladding. Alloys that are not resistant to acids and alkalis are subject to processing.
Clad steel coated with a metal layer is called biometal. It is protected from environmental factors, as well as exposure to organic acids. Titanium, nickel, stainless steel, silver, copper, gold, molybdenum can be used as protection.
After processing, the following changes are observed in the characteristics of the processed product:
- The hardness of the workpiece increases.
- The wear resistance indicator increases.
- The material is additionally protected from moisture, steam and organic acids.
However, this technological process also has disadvantages:
- After applying a protective coating using the hot rolling method, the price of the workpiece increases significantly.
- The clad layer on welds rusts many times faster.
Cladding is a “cheaper” way to protect materials from corrosion. The workpiece can be coated on different sides with different metals. For example, molybdenum is fixed on one side, and zinc on the other. In this way, it is possible to increase the wear resistance of the workpiece on the one hand and protect it from the destructive effects of acids on the other.
About large diameter pipes
The pipe industry keeps up with the times. Today, the priority is the production of risers used in the transportation of gas and oil on an industrial scale. Large diameter clad pipes have great practical application in this regard.
Therefore, Gazprom set a task for the domestic manufacturer: to launch BD clad pipes into mass production. Thanks to the implementation of such a project, domestic pipe makers will provide the gas industry with a new type of pipe products.
Clad types of BD pipes counteract carbon dioxide corrosion processes that are typical for oil and gas fields in the Far North and the shelf. They have excellent strength properties and the ability to withstand high pressures.
What and where is it used for?
Since clad metal is resistant to corrosion, it is used for the manufacture of parts in contact with liquids. Cladding is used to process parts that will experience increased mechanical stress. This is due to the fact that after applying a protective coating, the wear resistance indicator increases. In this way, you can protect mechanisms with cheap materials and save on purchasing solid products made from expensive metals or alloys.
Purchasing Tips
You can purchase high-quality gold-plated jewelry in jewelry stores, but you need to know some selection rules so as not to be mistaken in the value of the product.
Silver products must have a stamp confirming the hallmark . If it is not there, then it is not a precious metal, but a jewelry alloy coated with gold. Such products are often purchased because they are cheaper, but they quickly deform.
Different types of silver can be used to make jewelry, but experts recommend choosing 925 silver. Products made from it are of much higher quality, so they will last a long time. The percentage of gold plating should be more than 40%.
Peculiarities
The best material for processing is steel coated with a layer of aluminum. Using special equipment, the master applies a protective coating by spraying onto the work surface. This process is carried out using an oxygen burner. To increase the efficiency of the application method, it is necessary to use a plasmatron. The process of diffuse annealing helps to reliably fix the protective layer. During this process, the workpiece is subjected to heat treatment at temperatures up to 950 degrees Celsius. During this process, iron and aluminum atoms react and form a film that protects the surface from rust.
The thickness of the additional layer can range from 3 to 40% of the total thickness of the workpiece. The optimal thickness for protection from environmental factors is 0.15 mm.
Cladding is considered an additional process in metalworking, which allows a cost-effective way to protect the metal surface from corrosive destruction. A layer of metal is applied to both one and both sides of the workpiece. This allows you to increase the various characteristics of the working surfaces independently of each other.
Source of the article: https://metalloy.ru/obrabotka/zashhita/plakirovanie
Ways to do it at home
All products that do not have electrically conductive properties must be coated with graphite. The silver electroplating process, although very interesting, uses reagents that can cause harm to health, property and the environment. Therefore, before carrying out the procedure, you should choose the right place to ensure complete safety.
True, not all methods are like this. Let's look at popular ways to coat things with silver at home using electroplating.
Degreasing composition
Before starting the process, the products being processed must be degreased. To do this, you can use the recipe (values are based on 1 liter of water):
Heat the solution to 80...100 °C and hold for 15-60 minutes (until all contaminants are removed).
Chemical method
This type of silver coating at home consists of finishing with a photo fixative solution. Mode of application:
- mix hyposulfite with formaldehyde in proportions of 1 l/10 drops;
- 5 ml of ammonia is added;
- clean the item from grease and dirt;
- immerse in the finished substance for 1.5 hours;
- rinse, dry, wipe with soft fleece cloth.
The use of this method is considered gentle on health, since there is no strong release of chemical fumes. However, you should still take precautions to avoid getting a chemical burn.
Below watch a video about the chemical method of applying silver to products:
Using special pastes
Applying silver is a type of electroplating. The process does not require special equipment or work with hazardous chemicals, is suitable for products of any class, and the effect is achieved instantly. You can make a thick layer by performing the procedure 2-3 times:
- degrease the part;
- apply the paste thinly;
- dry;
- remove any remaining substance with water.
Permission is not required for purchase. The method is perfect for beginners, and the quality will not be inferior to similar procedures.
Silvering with heating of the workpiece
- Mix 100 g of argentum chloride with water;
- add 600 g of table salt and cream of tartar;
- Bring to a thick, homogeneous mass.
The resulting paste should be stored in a dark glass container.
- dilute the mixture (3 tbsp) with boiling water (5 l) in a copper container;
- place the item in the grill for 15-20 minutes.
This method of electroplating with silver will not add brightness. To get shine, you should use another recipe:
- boil water to 75...80 °C;
- mix the ingredients and add to the liquid;
- put an item;
- cook for 15 minutes.
Metal cladding
Modern manufacturers of pipes and pipeline fittings are seriously thinking about the need to extend the service life of their products. To achieve the desired result, it is necessary to use new materials created using innovative technologies. One of the most popular ways to improve quality characteristics is cladding. Metal cladding is the thermomechanical application of a thin protective layer of another metal or alloy to its surface. Sheets, plates, wire, and pipe products are coated in this way.
Cladding: concept and methods
Cladding is used to create a layer on the surface of a material that has certain properties. This may include hardness and wear resistance, as well as resistance to:
- the occurrence of corrosion;
- high temperatures;
- erosion.
In this way, you can not only create equipment and parts, but also restore them after wear.
The protective coating can be applied on one or both sides. Its thickness can range from a tenth to several millimeters (3-40% of the thickness of the metal to which it is applied). This method usually improves the quality characteristics of metals such as carbon and acid-resistant steels, titanium or copper alloys.
Which cladding method should I choose? The following methods are usually used:
- Batch rolling . Sheets of metal are collected and welded into a multilayer package. The number of layers depends on the purpose of the material and varies from 2 to 4. This method is used most often. It consists in the fact that the sheet and protective material are collected in a bag, which is sealed with sealed seams and heated to a temperature of 1450-1550K. Next, rolling is performed directly with a reduction value of more than 60%. This procedure results in welding of the metal and the cladding layer.
- Combined casting . Perforated separating sheets are installed into the ingot blank to determine the position of the future connection plane between the materials. After this, two metals are poured into the mold simultaneously. The specialist performing the work controls the equality of the heights of the liquid material. After this, the bimetallic ingot is rolled.
- Combination of liquid and solid metal . A solid slab is placed into an ingot blank and then liquid metal is poured around it. The layers are joined by rolling the resulting ingot.
- Explosion welding . The technique is used to create special-purpose products or connect pairs of materials that are difficult to clad in other ways. The explosion makes it possible to combine metals that do not dissolve in each other, form an intermetallic compound at elevated temperatures and differ in the level of resistance to deformation. The base and protective material is used in a cold state. An explosive with a detonator is placed on the surface of a metal cladding sheet located at an angle to the base. During the explosion, the plates collide and a jet of metal emerges from the surface of both layers. The deformation process occurs under a pressure difference from normal atmospheric pressure to 15 MPa (corresponding to the movement of shock wave fronts).
- Drawing of a pipe with a core . This method is used for cladding wire.
- Surfacing . This method is characterized by the absence of dilution of the working layer with the base metal. Methods for connecting layers can be different (forging, rolling, explosion). For cladding, the pressure welding method (or its variations) is used. Surfacing is accompanied by dilution of the protective layer material with the base.
Silvering. Silver cladding. FRAGET EPNS ALPACCA
Oh, how often a novice investor is scammed into purchasing an item positioned as solid silver, but in reality it turns out to be ordinary silver-plated metal. And how deceiving brands are, often everyone’s favorite “84” stands, suddenly overshadowing the mind, three-digit numbers per gram are already spinning in your head... and as a result, on your hands - “blanc metal” with a light “eaten” in a month with regular use ", a thin layer of silver.
In this case, these same numbers 90, 100, 120, 150, and there are also 13, 20, 40, 60, 80 and of course 84 - mean how many grams of pure silver were spent in alloying (plating with silver) 12 tablespoons and 12 forks This stamp began to be placed around 1850. This corresponds to 24 sq.dm. If we talk about a teapot, then this is how we count – 90 grams of pure silver per 24 sq.dm. area.
Numerical stamps on silver plated objects.
The production of silver-plated cutlery on an industrial level began in Germany in the mid-19th century. Two factors limited production: 1. Access to electricity was very limited at the time. 2. Electric current was quite weak compared to our time.
After some experimentation, the researchers found that the best results were achieved when they used a small plating bath, placing one dozen tablespoons and one dozen dinner forks in it and using 90 grams of pure silver and then depositing the silver until the silver anodes were used up and the silver settled firmly on the cutlery. This was time consuming and made the finished items quite expensive in the beginning. The engineers found that slightly more silver was deposited on the 12 spoons than half of the 90 grams used (because they have a larger surface area than a fork), slightly less than half was deposited on these 12 forks. Using a large bath would require a much longer metallization process, which would make the process even more expensive. Using more than 12 + 12 items in a bath would result in: 1. The items closest to the silver anodes would receive a much thicker plating. 2. The parts furthest from the anodes would receive very thin metallization. Thus, the thickness of the silver layer would differ significantly.
The use of 12 + 12 pieces as described above and 90 grams of pure silver became standard in Germany. The process was registered and these “90” began to be stamped on objects. If companies wanted to produce cheaper cutlery, they used less silver, 60 grams, 40 grams or even 20 grams, which made the plating very thin. Some used more, 100 or 150 grams. “60″, “40″, “20″, “100″, etc. were written on the objects, respectively.
The plating process was adapted to other cutlery items: knife handles, small spoons, etc., so that the silver layer on them was as thick as on table spoons and forks. Because the same standard process was used, they were all marked "90". New methods made it possible to coat more objects with silver in larger baths in a shorter time using large silver anodes. However, the thickness of the plating remained the same, so the mark remained the same.
As silver-plated cutlery became more available, numerous consumers began to wonder: how much pure silver did their cutlery actually contain? Manufacturers realized they could use the answer as a method to promote sales and began stamping another stamp that roughly reflected the actual grams of silver that coated the item. Unfortunately, they used two different systems: 1. Items that are usually made in dozens (dinner forks, knives, coffee spoons, etc.) are branded with the weight of silver used to plate the dozen pieces with a layer of silver. Thus, table forks and tablespoons were marked with a second mark of “45″, small objects were marked with a lower number (for example, “35″) since less silver was needed to provide them with the same thickness of plating. 2. Items that were usually purchased individually or in pairs were marked with a silver weight mark on the individual item.
Examples: If you have a tablespoon marked “90″ and “45″ this means: the standard process as described above was used on one spoon approximately 1/12 of 45 grams (approximately 3.75 grams) of pure silver were deposited. If you have a sugar scoop marked “90” and “2” this means: again the standard process was used, 2 grams of pure silver was used to coat the item. If you have a pair of salad cutters, each piece marked “90″ and “4″ this means: again the standard process was used on each piece, 4 grams of pure silver was used.
Why and where is cladding used?
Cladding is a universal technology that allows you to create or repair parts that are exposed to adverse environments during operation. It could be:
- various pollution;
- slag;
- steam;
- high temperatures.
Using this technique allows you to save money on purchasing new parts. The method makes it possible to obtain a bimetal (trimetal), which significantly extends the maintenance-free operation of pipelines and structures made from this material.
Mixing two metals allows you to avoid using products made from more expensive materials. Thanks to this feature, elements made using cladding began to be actively used in the construction of pipeline systems, gradually displacing products made from more expensive but less resistant to adverse influences materials.
Cladding technologies are actively used in jewelry. They allow you to create custom jewelry or more affordable products (for example, when applying gold plating to silver).
The method is also used in the production of coins.
Terms of use
Some buyers of gold-plated jewelry claim that the coating quickly wears off, and the products themselves lose their attractive and impressive appearance. For example, if a piece of jewelry quickly fades after contact with water, people think it is fake. But real gold-plated silver cannot deteriorate for this reason alone.
In order for products to delight you with their beautiful appearance for a long time, you should use them correctly:
- Earrings, chains and bracelets should be removed before showering or bathing. Jewelry becomes dull in soapy water, and frequent contact with it can easily damage the metal. If a person forgets to remove accessories, they should not be dried with a towel afterwards. Friction also negatively affects gold plating.
- When playing sports, it is not recommended to wear gold-plated silver as it deteriorates due to human sweat.
- Products should be stored in boxes or other closed containers that will protect them from sunlight and other negative factors. To prevent jewelry from scratching against each other, you should choose a separate box for each of them.
- Jewelry needs to be cleaned regularly to ensure it lasts a long time. But it is necessary to use certain methods of caring for gold-plated silver.
- Products should not be kept in a bathroom or kitchen area with high humidity levels, because the accessories will quickly lose their characteristic shine.
- If a person wears jewelry daily, he should wipe it with a suede cloth after each use. This material will help keep the product clean. In addition, suede will eliminate the need for radical methods of cleaning metal.
The thin layer of film covering silver can be damaged by negative factors. The quality of the base of the product also affects the coating itself. Other types of metal are added to the silver alloy. If there is too much nickel or copper in it, the jewelry will quickly turn black and become covered with plaque.
The process of metal oxidation can be accelerated by aggressive components included in cleaning products. Even some cosmetics can affect gold-plated jewelry. To avoid damage to accessories, they should not be worn in contact with chemicals.
Instead of a resume
Organizing the activities of modern production is not only the search for high-quality consumables and reliable sales, but also reducing the costs of creating products. That is why many enterprises that create metal parts and structures are increasingly giving preference to cladding. The material created using this technology is reliable, able to withstand heavy loads and last a much longer period of time. The area of application of bi- and trimetals is gradually expanding, and this is quite normal. The use of parts made of clad metals not only reduces the cost of creating a particular system, but also significantly increases its service life. When ordering such products, you should pay attention to the reputation of the manufacturing company, the availability of a warranty and the possibility of obtaining all technical documentation.
Contents: Classification of devices Recommendations when selecting an electric drive Ball valves are a type of pipeline fittings, the locking (regulating) element of which is made in the shape of a sphere. They are actively used in the construction of oil...
Contents: Areas of application Advantages of steel bends Classification The construction of pipelines of any purpose and complexity is not complete without the use of elements such as bends. These are connecting parts that allow…
Source of the article: https://rgsnab.com/articles/plakirovanie-metallov/
What determines the quality and service life of gold coating?
In our country, when plating products with gold, high-grade metal is always used, above 750 fineness, but most often - 999. It is somewhat less common to find plating with gold of 14 carats, or 585 fineness. Abroad, both 12-carat gold and lower, 8-9 carats, are used for this purpose. This factor has a significant impact on the strength of the coating and service life. In addition, depending on the sample, the coating may have different colors.
Gold Plated Silver Stud Earrings
Experts believe that the main condition for high-quality gilding is that the silver on which the coating is applied must have a purity of at least 925, and the applied gold must be at least 42%. The thickness of the applied gold should be more than 2.5 microns, and the fineness should be 999. Under such conditions, the gold plating will last a long time and reliably.