Global production of primary and secondary refined copper is currently around 15-16 million tonnes per year. Half of all copper produced is used in the electrical industry. The bulk of copper is obtained from primary raw materials, but a very significant proportion of copper is also produced from secondary raw materials. Thus, M1 sheet and electrical copper tapes, copper rods, and wire are produced mainly from primary raw materials; from the secondary - copper tube M2, again wire, roofing copper.
Where is copper ore mined?
The main deposits of copper ores in Russia are concentrated in the Urals, eastern Siberia and the North Caucasus. The list of the main Ural deposits located in the Sverdlovsk region includes Kirovogradskoye, Revdinskoye, Degtyarskoye and Krasnouralskoye.
Interesting materials:
Why was the VChK created? Why set the table beautifully? Why Ski Socks? Why is there an Anvil on the Secateurs? Why do you cover garlic for the winter? Why do you need a block launcher? Why is friGate needed? Why is karate needed? Why is biology needed in the modern world? Why are jellyfish needed in nature?
COPPER PRODUCTION METHODS
There are two known methods for extracting copper from ores and concentrates: hydrometallurgical and pyrometallurgical. The first of them has not found widespread use. It is used in the processing of low-grade oxidized and native ores. This method, unlike the pyrometallurgical method, does not allow the extraction of precious metals along with copper. The second method is suitable for processing all ores and is especially effective when the ores are subjected to beneficiation.
PYROMETALLURGICAL METHOD FOR COPPER PRODUCTION
The basis of the pyrometallurgical process is smelting, in which the molten mass is divided into two liquid layers: matte-alloy of sulfides and slag-alloy of oxides. The smelting process involves either copper ore or roasted copper ore concentrates. Roasting of concentrates is carried out in order to reduce the sulfur content to optimal values. Liquid matte is blown in converters with air to oxidize iron sulfide, convert iron into slag and separate blister copper. The blister copper is then subjected to refining – purification from impurities.
Copper ore beneficiation
Most copper ores are enriched by flotation. The result is a copper concentrate containing 8-35% Cu, 40-50% S, 30-35% Fe and waste rock, the main components of which are SiO2, Al2O3 and CaO. The concentrates are usually fired in an oxidizing environment to remove about 50% of the sulfur and produce a fired concentrate with the sulfur content necessary to produce a sufficiently rich matte when smelted. Firing ensures good mixing of all components of the charge and heating it to 550-600 ºС and, ultimately, reducing fuel consumption in a reverberatory furnace by half. However, when remelting the fired charge, copper loss in the slag and dust entrainment increase slightly. Therefore, usually rich copper concentrates (25-35% Cu) are smelted without firing, and poor copper concentrates (8-25% Cu) are fired. Multi-hearth furnaces with mechanical overheating are used for firing concentrates. Such furnaces operate continuously.
Copper matte smelting
Copper matte, consisting mainly of copper and iron sulfides (Cu2S+FeS=80-90%) and other sulfides, as well as oxides of iron, silicon, aluminum and calcium, is smelted in various types of furnaces. It is advisable to enrich complex ores containing gold, silver, selenium and tellurium so that not only copper, but also these metals are converted into concentrate. The concentrate is melted into matte in reverberatory or electric furnaces. It is advisable to process sulphurous, pure copper ores in shaft furnaces. When the sulfur content in ores is high, it is advisable to use the so-called copper-sulfur smelting process in a shaft furnace with gas capture and extraction of elemental sulfur from them. Copper ore, limestone, coke and recycled products are loaded into the furnace. Loading is carried out in separate portions of raw materials and coke. A reducing environment is created in the upper horizons of the mine, and an oxidizing environment is created in the lower part of the furnace. The lower layers of the charge melt, and it gradually falls down towards the flow of hot gases. The temperature at the tuyeres reaches 1500 ºС; at the top of the furnace it is approximately 450 ºС. Such a high temperature of the exhaust gases is necessary in order to ensure the possibility of cleaning from dust before the condensation of sulfur vapor begins. In the lower part of the furnace, mainly at the tuyeres, the following main processes take place: a) Combustion of coke carbon C + O2 = CO2 b) Combustion of sulfur in the form of iron sulfide 2FeS + 3O2 = 2FeO + 2SO2 c) Formation of iron silicate 2 FeO + SiO2 = (FeO)2 × SiO2 Gases containing CO2, SO2, excess oxygen and nitrogen flow upward through the charge column. Along this gas path, heat exchange occurs between the charge and them, as well as the interaction of CO2 with the carbon of the charge. At high temperatures, CO2 and SO2 are reduced by coke carbon and thus form carbon monoxide, carbon disulfide and carbon sulfur monoxide (COS): CO2 + C = 2CO 2SO2 + 5C = 4CO+ CS2 SO2 + 2C = COS+ CO In the upper horizons of the furnace, pyrite decomposes according to the reaction : FeS2 = Fe + S2 At a temperature of about 1000 0, the most fusible eutectics of FeS and Cu2S melt, resulting in the formation of a porous mass. In the pores of this mass, the molten flow of sulfides meets the ascending flow of hot gases and at the same time chemical reactions occur, the most important of which are listed below: a) the formation of copper sulfide from copper oxide 2Cu2O + 2FeS + SiO2 = (FeO)2 × SiO2 + 2Cu2S; b) formation of silicates from iron oxides 3Fe2O3+ FeS + 3.5SiO2 = 3.5(2FeO × SiO2) + SO2; 3Fe3O4+ FeS + 5SiO2 = 5(2FeO × SiO2) + SO2; c) decomposition of CaCO3 and formation of lime silicate CaCO3 + SiO2= CaO × SiO2 + CO2; d) reduction of sulfur dioxide to elemental sulfur SO2 + C = CO2+ ½ S2 As a result of smelting, matte containing 8-15% copper (Cu), slag consisting mainly of iron and lime silicates, top gas containing sulfur and its compounds ( S2, COS, H2S), and carbon dioxide (CO2). Dust is first deposited from the gas, then sulfur is extracted from it (up to 80% S). To increase the copper content of the matte, it is subjected to contractile smelting. Melting is carried out in the same shaft furnaces. The matte is loaded in pieces of 30-100 mm in size along with quartz flux, limestone and coke. Coke consumption is 7-8% of the charge weight. As a result, copper-enriched matte (25-40% Cu) and slag (0.4-0.8% Cu) are obtained. Melting point for remelting concentrates, as already mentioned, reverberatory and electric furnaces are used. Sometimes kilns are located directly above the platform of reverberatory kilns so as not to cool the calcined concentrates and use their heat. As the furnace charge heats up, the following reduction reactions of copper oxide and higher iron oxides occur: 6CuO + FeS = 3Cu2O+ SO2 + FeO; FeS + 3Fe3O4+ 5SiO2 = 5(2FeO × SiO2) + SO2 As a result of the reaction of the resulting copper oxide Cu2Oс FeS, Cu2S is obtained: Cu2O + FeS = Cu2S+ FeO Copper and iron sulfides, fusing with each other, form the primary matte, and molten iron silicates, flowing down the slope surfaces, dissolve other oxides and form slag. Noble metals (gold and silver) dissolve poorly in slag and almost completely turn into matte. Reflective smelting matte consists of 80-90% (by weight) of copper and iron sulfides. Matte contains, %: 15-55 copper; 15-50 iron; 20-30 sulfur; 0.5-1.5 SiO2; 0.5-3.0 Al2O3; 0.5-2.0(CaO + MgO); about 2% Zn and small amounts of gold and silver. The slag consists mainly of SiO2, FeO, CaO, Al2O3 and contains 0.1-0.5% copper. The extraction of copper and precious metals into matte reaches 96-99%.
Copper matte conversion
The Russian engineer G. S. Semennikov invented a converter for producing copper in 1866. He proposed using a Bessemer-type converter for blowing matte. Blowing the matte with air from below ensured the production of only semi-sulfur copper (about 79% copper) - the so-called white matte. Further blowing led to solidification of the copper. In 1880, a Russian engineer proposed a converter for blowing matte with side blowing, which made it possible to obtain blister copper in converters. The converter is made 6-10 meters long, with an outer diameter of 3-4 meters. The productivity per operation is 80-100 tons. The converter is lined with magnesite bricks. The molten matte is poured and the products are drained through the neck of the converter, located in the middle part of its body. Gases are removed through the same neck. Lances for air injection are located along the generatrix surface of the converter. The number of tuyeres is usually 46-52, and the tuyere diameter is 50mm. Air flow reaches 800 m2/min. Matte is poured into the converter and quartz flux containing 70-80% SiO2 and usually a certain amount of gold is fed. It is fed during melting using pneumatic loading through a round hole in the end wall of the converters, or it is loaded through the neck of the converter. The conversion process is carried out in a cyclic mode at a temperature of 1250-1350 °C and consists of two periods. In the first period, iron and associated sulfur are removed. The first period (oxidation of iron sulfide to produce white matte) lasts from 6 to 24 hours depending on the copper content in the matte. Loading of quartz flux begins from the beginning of purging. As slag accumulates, it is partially removed and a new portion of the original matte is poured into the converter, maintaining a certain level of matte in the converter. In the first period, the following sulfide oxidation reactions occur: 2FeS + 3O2 = 2FeO + 2SO2 + 930360 J 2Cu2S + 3O2 = 2Cu2O + 2SO2 + 765600 J While FeS exists, cuprous oxide is unstable and turns into sulfide: Cu2O + FeS = Cu2S+ FeO Iron oxide is slaged by quartz flux added to the converter: 2FeO + SiO2 = (FeO) × SiO2 If there is a lack of SiO2, ferrous oxide is oxidized by magnetite: 6FeO + O2 = 2Fe3O4, which turns into slag. As a result of these exothermic reactions, the temperature of the matte being poured increases from 1100–1200 to 1250–1350 0C. A higher temperature is undesirable, and therefore, when blowing through poor mattes containing a lot of FeS, coolants are added - solid matte, copper splashes, crusts from ladles, copper concentrates. As a result, mainly the so-called white matte, consisting of copper sulfides, remains in the converter, and the slag is drained during the smelting process. It consists mainly of various oxides of iron (magnetite, ferric oxide) and silica, as well as small quantities of alumina, calcium oxide and magnesium oxide. In this case, as follows from the above, the content of magnetite in the slag is determined by the content of silica. 1.8-3.0% copper remains in the slag. To extract it, the slag in liquid form is sent to a reverberatory furnace or to the hearth of a shaft furnace. In the second period, called the reaction period, which lasts 2-3 hours, blister copper is formed from white matte. During this period, copper sulfide is oxidized and copper is released by the exchange reaction: 2Cu2S + 3O2 = 2Cu2O + 2SO2 Cu2S + 2Cu2O = 6Cu + O2 Thus, as a result of blowing, blister copper is obtained containing 98.4-99.4% copper, 0 .01-0.04% iron, 0.02-0.1% sulfur, and a small amount of nickel, tin, arsenic, silver, gold and converter slag containing 22-30% SiO2, 47-70% FeO, about 3 % Al2O3 and 1.5-2.5% copper.
Answers to exercises § 7. Chemistry 9th grade.
| Exercise 1 In the work of the German scientist in the field of metallurgy and physician G. Agricola (16th century) “12 books on metals” it is said: “By subjecting the ore to heating, roasting and calcination, this removes part of the substances mixed with the metal...” and further “... smelting is necessary , since only through it are rocks and hardened juices (brines) separated from metals, which acquire their characteristic color, are purified and become useful to man in many ways.” What types of metallurgy did Agricola write about? Illustrate his statement with examples of chemical reaction equations. Pyrometallurgy roasting: 2CuS + 3O2 = 2CuO + 2SO2 smelting: 2CuO + C = 2Cu + CO2 | Exercise 2 Which method of producing copper - using sulfuric acid or bacterial - is more environmentally friendly? The bacterial method is safer from an environmental point of view, since the process of obtaining copper does not use aggressive, harmful substances and therefore produces less waste harmful to the environment | Exercise: 3 Why can’t alkali and alkaline earth metals be obtained by the hydrometallurgical method? The hydrometallurgical method is the recovery of metals from aqueous solutions of their compounds (soluble salts of these metals). Since alkali and alkaline earth metals interact with water, it is impossible to obtain them from an aqueous solution of their salt; they react with water. | Exercise: 4 Propose a technological chain for the production of lead from the galena mineral PbS. Write down the reaction equations. PbS → PbO → Pb 2PbS + O2 = 2PbO + 3SO2 PbO + Al = Pb + Al2O3 |
reshebnikxim.narod.ru
Technology
Bessemerizing is an iron smelting process that produces steel of relatively high quality. It should be noted that such technology is used extremely rarely today. This is due to the emergence of quite a large number of modern technologies that make it possible to obtain higher quality steel in less time.
The entire Bessemer steel production process can be divided into several main stages:
- Cast iron is poured into the converter through the neck. An important point is that in this position the device must be in a horizontal position, since there is a possibility that the nozzle will be filled with metal. Nozzles are necessary to blow through the charge. It is the oxidation of impurities and their removal as slag that makes it possible to obtain steel of improved quality.
- The next stage is to start the blast and turn the converter into a vertical position.
- In order to ensure the oxidation of harmful impurities and excess carbon, the metal is blown with air. At this stage, slag is formed, with which unnecessary chemicals are removed.
- After a sufficiently long period of purging, the converter is turned over again into a horizontal position, and the purging of molten metal stops.
- The molten metal is drained into a ladle and deoxidized by adding special substances.
At the time of the start of purging the composition, active oxidation of manganese and silicon occurs. At the initial stage, carbon is practically not oxidized. This is due to the fact that this component reacts exclusively to exposure to high temperatures. In addition, the process of oxidation of impurities is influenced by thermodynamic factors that determine the activity of oxygen transfer to the sites of the Bessemer process.
Considering this technology, we note the following points:
- At the first stage, a large number of different slags are formed, which contains a high concentration of silica. The time interval for the first stage is 2-5 minutes.
- The second stage of the Bessemer production process provides the most favorable conditions for carbon oxidation. An example is an increase in operating temperature to approximately 2000 degrees Celsius. The duration of this stage is approximately 13 minutes. At the end of this stage, the temperature drops to approximately 1600 degrees Celsius.
- High quality steel can be achieved using various bessemerization methods. It all depends on the characteristics of the composition of the scrap used and the concentration of the cream in the composition.
- In order to eliminate the possibility of metal overblowing, the active air supply is stopped already at the second stage.
- Only at the third stage can active oxidation of iron be noted, which causes the release of brown smoke. This stage begins at the moment when the carbon concentration is less than 0.1%.
As previously noted, the Bessemer method of steel production has become widespread due to its high productivity. Foundries quite often install equipment that has a charge of about 35 tons.
Bessemer method of steel smelting
Today, the Bessemer method of steel production is practically not used, which is due to the low quality of the resulting metal and its fairly high cost.
Source
§ 12. Obtaining metals -
1. In the work of the German scientist in the field of metallurgy and physician G. Agricola (16th century) “12 books on metals” it is said: “By subjecting the ore to heating, roasting and calcination, this removes part of the substances mixed with the metal...” and further “... smelting is necessary, since only through it are rocks and solidified juices (brines) separated from metals, which acquire their characteristic color, are purified and become useful to man in many ways.”
What types of metallurgy did Agricola write about? Illustrate his statement with examples of chemical reaction equations. G. Afikol had pyrometallurgy in mind in his book.
Pyrometallurgical processes involve roasting, in which metal compounds contained in ores, in particular sulfides, are converted into oxides, and sulfur is removed in the form of SO2, for example: 2. Which method of obtaining copper - using sulfuric acid or bacterial - is environmentally safer?
The bacterial method of producing copper is environmentally safer.
3. Why can’t alkali and alkaline earth metals be obtained by the hydrometallurgical method?
Alkali and alkaline earth metals cannot be obtained by the hydrometallurgical method, because this method is based on the separation of metals from solution under the influence of an electric current.
And if we isolate an alkali metal in its pure form from a solution, it will immediately react with water, forming a hydroxide. 4. Propose a technological chain for the production of lead from the galena mineral PbS. Write down the reaction equations. 5. How can iron and sulfuric acid be obtained from FeS2 pyrite? Write down the reaction equations. 6. How many kilograms of copper are obtained from 120 tons of enriched rock containing 20% copper (I) sulfide if the copper yield is 90% of the theoretically possible?
gdz-himiya.ru
Areas of use
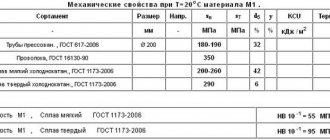
Copper is in greatest demand in electrical engineering. Due to its low electrical resistance, it is actively used for the manufacture of wires, cables, conductive elements of transformers, and electric motor windings are made from it. More than half of all mined copper is consumed by the industry.
The metal has good strength and ductility, which makes it suitable for the manufacture of products of complex shapes: household items, jewelry; for creating seamless pipes. Copper pipes are often used in equipment and various engineering systems to organize heat exchange. The properties of copper such as low coefficient of thermal expansion and high thermal conductivity are used here.
Copper alloys are widely used:
- bronze - a compound with tin, the name is also used for alloys of copper with other metals, such as aluminum, lead, silicon, beryllium and others;
- brass - with zinc;
- cupronickel, nickel silver, manganin - with nickel;
Copper is also included as one of the components in other compounds, for example, in duralumin. Jewelers often use copper alloys with the addition of gold.