Optical emission spectral analysis of C, S, P.
Optical emission spectrometers are universal instruments that can solve a wide range of analytical problems. Their work is based on the principles of atomic emission spectral analysis of the elemental composition of matter:
- the spectrum of excited atoms and ions is individual for each element;
- the intensity of the spectral line depends on the concentration of the element in the test sample.
Emission spectral devices are widely used in metallurgy, due to the following advantages of the method:
- Possibility of studying samples in different states of aggregation.
- The analysis is non-destructive.
- The number of elements studied is practically unlimited. These include carbon, sulfur and phosphorus, which are of particular interest to metallurgists.
- To conduct a test, a small amount of a substance is sufficient as a sample.
- High sensitivity and accuracy.
- Expressiveness.
- Possibility of conducting certification analysis.
To analyze carbon, sulfur and phosphorus using emission spectrometers, certain conditions must be created in the device, namely: an oxygen-free atmosphere. Otherwise, it is not possible to determine elements whose wavelength is shorter than 185 nm. Currently, oxygen removal in the device is carried out in two ways:
- by pumping with inert gas;
- vacuuming.
Each deoxygenation system has certain operating and maintenance features, so when choosing a device for analyzing carbon, sulfur and phosphorus, their advantages and disadvantages should be taken into account. This will allow you to select a spectrometer that optimally matches the analytical task, the requirements for the accuracy of research results, and has satisfactory economic indicators.
Optical emission devices involving pumping with inert gas
Argon is most often used in spectral instruments for deoxygenation. To remove oxygen, one of the following systems is provided:
- Open. As a result of purging, oxygen is displaced, and inert gas is removed from the device into the surrounding atmosphere.
- Closed. When the inert gas passes through, oxygen is captured, which is subsequently purified using a filter. The gas continues to move through a closed system, the pressure in which is provided by the pump.
Devices with an open deoxygenation system are characterized by simple design and lower cost. However, in this case, the degree of purification is at a low level, and argon is consumed irrevocably. The use of such spectrometers is advisable when requirements for analytical characteristics are reduced, both on the part of the consumer and on the part of the manufacturer.
The design of devices with a closed deacidification system becomes more complicated, since additional components and their maintenance are required to ensure functionality:
- Pump with power supply.
- Gas cylinder to compensate for losses.
- Additional filter element.
Each of these instrument components requires maintenance and consumables require replacement, which is associated with additional costs. In addition, as a result of unprofessional actions of maintenance personnel, there is a risk of air in the system when replacing the filter. Eliminating the consequences of this requires not only additional material costs, but also time.
Optical emission devices with a vacuum system
The vacuum system allows to obtain a low residual oxygen concentration, which is many times lower than in an open deoxygenation system and comparable to the best results obtained in closed ones. It should be noted that there is no need to use inert gas.
This oxygen removal system is used in the most advanced spectral instruments. They are equipped with an oil pump, which is complemented by special oil traps. In addition, a valve is provided that, in the event of an emergency power outage, prevents damage to the spectrometer by oil as a result of its penetration into the vacuum line.
Two-stage oil foreline pumps are the most preferred equipment compared to oil-free diaphragm models. They have a comparable cost, but at the same time are tens of times superior to the latter in terms of oxygen removal, and also have a significant service life and are much easier to maintain.
Universal desktop and stationary spectrometers Iskroline 100/300 are excellent examples of devices in which a vacuum system is implemented to remove oxygen. They are capable of determining more than 70 elements, which include carbon, sulfur and phosphorus, with a detection limit of up to 0.0001%. The devices allow for quick and accurate spectral analysis of steels, and are characterized by high spectral resolution, high accuracy of measurement results and high quality manufacturing.
The influence of chemical composition on the mechanical properties of steel
Each chemical element that makes up steel affects its mechanical properties in its own way - improves or worsens.
Carbon (C), which is an essential element and is usually found in steel in the form of the chemical compound Fe3C (iron carbide), increases its content to 1.2%, increases the hardness, strength and elasticity of steel and reduces toughness and weldability. This also reduces machinability and weldability.
Silicon (Si) is considered a beneficial impurity and is introduced as an active deoxidizer. As a rule, it is contained in steel in small quantities (up to 0.4%) and does not have a noticeable effect on its properties. But when the silicon content is more than 2%, the steel becomes brittle and breaks during forging.
Manganese (Mn) is contained in ordinary carbon steel in small quantities (0.3-0.8%) and does not have a serious effect on its properties. Manganese reduces the harmful effects of oxygen and sulfur, increases the hardness and strength of steel, its cutting properties, increases hardenability, but reduces resistance to impact loads.
Sulfur (S) and phosphorus (P) are harmful impurities. Their content, even in small quantities, has a harmful effect on the mechanical properties of steel. A steel content of more than 0.045% sulfur makes the steel red-brittle, i.e. one that cracks when forged in a heated state. Steel is protected from red brittleness by manganese, which binds sulfur into sulfides (MnS). The content of phosphorus in steel is more than 0.045%, making the steel cold-brittle, i.e. easily broken when cold. Phosphorus somewhat improves the machinability of steel, as it promotes chip separation.
Niobium (Nb) improves the acid resistance of steel and helps reduce corrosion in welded structures.
Titanium (Ti) increases the strength, density and ductility of steel, improves machinability and corrosion resistance. Increases the hardenability of steel at low contents and decreases at high contents.
Chromium (Cr) increases strength, hardenability and heat resistance, cutting properties and abrasion resistance, but reduces the toughness and thermal conductivity of steel. The content of a large amount of chromium (in ordinary types of steel reaches 2%, and in special types - up to 25%) makes the steel stainless and ensures the stability of magnetic forces.
Molybdenum (Mo) increases the strength characteristics of steel, increases hardness, red resistance, and anti-corrosion properties. Makes it heat-resistant, increases the load-bearing capacity of structures under shock loads and high temperatures. It makes welding difficult because it actively oxidizes and burns out.
Nickel (Ni) increases toughness, strength and elasticity, but slightly reduces the thermal conductivity of steel. Nickel steels are easy to forge. The significant nickel content makes the steel non-magnetic, corrosion-resistant and heat-resistant.
Tungsten (W), forming solid chemical compounds in steel - carbides, sharply increases hardness and red-hardness. Increases the performance of steel at high temperatures, its hardenability, increases the resistance of steel to corrosion and abrasion, and reduces weldability.
Vanadium (V) provides fine-grained steel, increases hardness and strength. Increases the density of steel, as it is a good deoxidizer. Reduces the sensitivity of steel to overheating and improves weldability.
Cobalt (Co) increases heat resistance, magnetic properties, and increases impact resistance.
Aluminum (Al) is an active deoxidizer. Makes steel fine-grained, homogeneous in chemical composition, prevents aging, improves stampability, increases hardness and strength, and increases oxidation resistance at high temperatures.
Copper (Cu) increases corrosion resistance, yield strength and hardenability. Does not affect weldability.
For a comprehensive understanding and analysis of the processes occurring during alloying and deformation of steels, knowledge of the relationships between the chemical composition and mechanical properties plays an important role.
The purpose of this research is to study the complex effect of chemical composition on the yield strength σT of reinforcing steel of class A500C
.
During September and October of this year, samples of reinforcing bars with a diameter from Ø16 to Ø36 were tested in the Laboratory for Testing Building Materials and Structures of the State Budgetary Institution “TsEIIS”. More than 30 parallel tests were performed. At the same time, for the same sample of a given standard size of reinforcing bars, the actual mass fraction of chemical elements was determined using a PMI-MASTER SORT optical emission spectrometer (Fig. 1) and the mechanical properties of steel using an IR-1000M-auto testing machine (Fig. 2 ).
Fig. 1 - Testing a reinforcing bar to determine the chemical composition of steel.
Fig. 2 - Tensile testing of reinforcing steel.
To ensure the reliability of statistical conclusions and meaningful interpretation of research results, the required sample size was first determined, i.e. minimum number of parallel tests. Since in this case the tests are carried out to assess the mathematical expectation, then with a normal distribution of the value under study, the minimum required volume of tests can be found from the relationship:
where υ is the sample coefficient of variation,
tα,k – Student coefficient,
α=1-P – significance level (P – confidence probability),
k = n-1 – number of degrees of freedom,
ΔМ – maximum relative error (tolerance) when estimating the mathematical expectation in fractions of the mathematical expectation (ΔМ = γ*δМ, where γ is the general coefficient of variation, δМ is the maximum error when estimating the mathematical expectation in fractions of the standard deviation).
As a rule, the general coefficient of variation γ is unknown, and it is replaced by the sample coefficient of variation υ, to determine which we conducted a series of ten preliminary tests.
Based on the results of the tests and calculations performed with a confidence probability of P = 0.95, the required sample size was obtained equal to n = 26
. The actual number of tests, as mentioned above, was 36.
The array of data obtained from the results of parallel tests was processed using multivariate correlation analysis.
The multiple regression equation can be represented as:
Y = f (β, X) + ε,
where X=(X1, X2,…, Xm) – vector of independent (initial) variables; β – vector of parameters (to be determined); ε – random error (deviation); Y – dependent (calculated) variable.
The development of a multiple correlation model is always associated with the selection of significant factors that have the greatest impact on the outcome trait. In our case, three elements (Al, Ti, W) were excluded from further consideration due to their low mass fraction (
If you find an error: select the text and press Ctrl+Enter
Useful and special impurities in steel
Steel contains harmful and beneficial impurities. First, let's look at the useful ones, which include manganese and silicon:
- Manganese is a chemical element that increases the hardenability of steel and reduces the influence of sulfur, which has a harmful effect on the metal.
- Silicon - an admixture of this element helps to deoxidize steel and, as a result, increase its strength. It is specially added to the metal during its smelting.
Carbon steel contains an admixture of silicon of no more than 0.35–0.4% and manganese in an amount of 0.5–0.8%. The transition of manganese and silicon into steel occurs during deoxidation during smelting. These chemical elements combine with the oxygen of iron oxide FeO, and then, turning into oxides, pass into slag, that is, in other words, they deoxidize the steel.
This process has a beneficial effect on the properties of steel. Due to degassing of the metal by silicon, its density increases. Part of the chemical element remains in the ferrite (solid solution) after deoxidation, which leads to a significant increase in the yield strength. At the same time, the ability of steel to cold heading and drawing is reduced.
For this reason, manufacturers are reducing the amount of silicon in steels produced for cold forming and heading. The strength of the metal is significantly increased due to the admixture of manganese. The latter greatly reduces the red brittleness of steel, leaving ductility almost unchanged. Thus, the brittleness of steel sharply decreases when exposed to high temperatures, which arose due to the presence of sulfur.
To obtain steels with certain properties, special impurities are added to the metal. They are called alloying elements. Steels are called alloyed steels.
Let us dwell in detail on the purpose of some elements:
- Aluminum - its admixture helps to increase the scale and heat resistance of steel.
- Copper – increases the resistance of steel to corrosion.
- Chromium – increases the strength and hardness of steels, increases resistance to corrosion, while ductility decreases slightly. What makes stainless steel stainless is its high chromium content.
- Nickel – increases ductility, strength, and makes steel resistant to corrosion.
- Tungsten – when added to steel, creates corbides (chemical compounds of increased hardness). They significantly increase red resistance and hardness. Under the influence of tungsten, steel stops expanding during heating, and its brittleness disappears when tempered.
- Vanadium – contributes to an increase in the density, strength and hardness of steel. It is recognized as an excellent deoxidizing agent.
- Cobalt - under its influence, heat resistance, resistance to shock loads increases, and magnetic properties increase.
- Molybdenum – steel’s resistance to oxidation improves when exposed to high temperatures, elasticity and red-hardness increases, corrosion resistance increases, and tensile strength increases.
- Titanium - being an excellent deoxidizer, it increases resistance to corrosion, increases the density and strength of the metal, and improves its workability.
- Cerium – helps to increase the ductility and strength of steel.
- Zirconium (Z) - affects the grain size of steel, making it possible to produce metal with a specified grain size, making it finer.
- Lanthanum, neodymium and cesium - reduce the porosity of steel, reduce the amount of sulfur, make the surface quality better and the grain finer.
Impurities: permanent, hidden and random
Manganese, silicon, aluminum, sulfur and phosphorus
classified as
permanent impurities
.
Aluminum, together with manganese and silicon, is used as a deoxidizer and therefore they are always present in small quantities in deoxidized steels. Iron ores, as well as fuels and fluxes, always contain a certain amount of phosphorus and sulfur, which remain in cast iron and then pass into steel
.
Nitrogen
called
a hidden
impurity - it enters steel mainly from the air.
To random
impurities include
copper, arsenic, tin, zinc, antimony, lead
and other elements. They end up in the steel with the charge - with ores from various deposits, as well as from iron scrap.
All impurities - permanent, hidden and accidental - are inevitable to varying degrees due to steel production technology. Thus, mild steel usually contains these impurities within the following limits: 0.3-0.7% manganese; 0.2-0.4% silicon; 0.01-0.02% aluminum; 0.01-0.05% phosphorus, 0.01-0.04% sulfur, 0.-0.2% copper. In these quantities, these elements are considered as impurities, and in larger quantities, which are intentionally added to steel, they are already considered alloying elements.
The influence of carbon and permanent impurities on the properties of steel
Carbon steel is a multicomponent alloy containing, in addition to iron and carbon, a number of permanent or inevitable impurities: Mn, Si, S, P, O, N, H, etc.
which influence its properties. These same impurities, only in larger quantities, are also found in cast iron.
The structure of steel, after slow cooling, consists of ferrite and cementite. The amount of cementite in steel increases in direct proportion to the carbon content. For example, at 0.37% C - C -5%, at 0.7% C -10% at 2% - 30% C
With an increase in carbon in steel, hardness, tensile strength, and yield strength increase, but relative elongation, relative contraction, and specific viscosity decrease.
When the carbon content in steel is more than 1%, its hardness increases and the tensile strength decreases. This is explained by the precipitation of secondary cementite along the boundaries of the former austenite grain, forming a continuous network. During tensile tests, high stresses arise in this mesh and cementite, being brittle, is destroyed.
Increasing the carbon content above 0.4% and decreasing below 0.3% leads to deterioration in machinability.
An increase in carbon content reduces the technological ductility of steel during hot and especially cold working and impairs weldability.
The silicon content as an impurity is usually up to 0,4%
, manganese – up to
0,5-0,8%
.
Manganese and silicon are transferred into steel during its deoxidation during smelting. They deoxidize steel, that is, with oxygen, iron oxide FeO, in the form of oxides they pass into slag. Deoxidation improves the properties of steels. Silicon, by degassing the metal, increases the density of the ingot.
Silicon greatly increases the yield strength of steel.
Manganese increases the strength of steel without reducing its ductility.
Manganese reduces the harmful effects of sulfur and oxygen.
The influence of phosphorus on the properties of steels
Phosphorus (P) segregates during steel solidification, but to a lesser extent than carbon and sulfur. Phosphorus dissolves in ferrite and thereby increases the strength of steels. As the phosphorus content in steels increases, their ductility and toughness decrease and their tendency to cold brittleness increases.
The solubility of phosphorus at high temperatures reaches 1.2%. With decreasing temperature, the solubility of phosphorus in iron drops sharply to 0.02-0.03%. This amount of phosphorus is typical for steels, that is, all phosphorus is usually dissolved in alpha iron.
Phosphorus has a strong tendency to segregate at grain boundaries, resulting in temper brittleness in alloy steels, especially in manganese, chromium, magnesium-silicon, chromium-nickel and chromium-manganese steels. Phosphorus, in addition, increases the hardening of steels and, like silicon, slows down the decomposition of martensite in steels.
Increased phosphorus content is often specified in low-alloy steels to improve their machining, especially automatic machining.
In low-alloy structural steels with a carbon content of about 0.1%, phosphorus increases strength and resistance to atmospheric corrosion.
In austenitic chromium-nickel steels, phosphorus additions help increase the yield strength. In strong oxidizers, the presence of phosphorus in austenitic stainless steels can lead to grain boundary corrosion. This is due to the phenomenon of phosphorus segregation along grain boundaries.
Theoretical foundations of metallurgical production
| 14.1 The role of sulfur in steelmaking processes |
Sulfur is one of the most harmful impurities in steel, reducing the mechanical strength, ductility and weldability of the metal, its performance properties, etc. Only a few examples of the negative impact of sulfur on the quality of the metal will be considered below.
Sulfur has unlimited solubility in liquid iron. There is evidence that up to 38% of sulfur can be dissolved in iron when its content in pure FeS sulfide is 36.5%. When a metal transitions from a liquid to a solid state, the solubility of sulfur in it decreases sharply and continues to decrease when the solid metal is cooled from the crystallization temperature to room temperature. So, for example, in -Fe at 1365°C sulfur dissolves in an amount of ~ 0.055%, in -Fe at 910°C ~ 0.015%, and at room temperature 0.001 – 0.002%. Thus, when the metal solidifies and cools, almost the entire amount of sulfur contained in the melt is released from the supersaturated solution in the form of iron sulfide FeS or oxysulfide eutectic.
From the phase diagram of the Fe-S system it follows that the melting point of pure iron sulfide is 1190°C, and the oxysulfide melt has a eutectic with a solidification temperature of ~ 985°C, which is significantly lower than the melting point of the metal. Therefore, during the crystallization of steel, inclusions of iron sulfide and oxysulfides are released in liquid form. Wetting the metal well, non-metallic inclusions in the form of thin films are located at the grain boundaries of the cast metal, occupying a large area.
At temperatures above 1000°C (red hot temperature), these inclusions are in a liquid or plastic state, which weakens the bond between the grains of the metal and its ability to undergo hot plastic deformation. This phenomenon is called red brittleness of steel. Red brittleness causes: the formation of hot cracks on the surface of ingots, cast billets and products; the appearance of flaws, cracks and other surface defects during rolling; poor welding of the internal voids of the metal during rolling, as a result of which the head trim from the ingots increases with increasing sulfur concentration, etc.
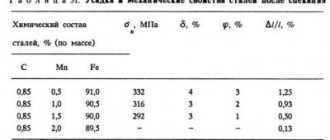
Currently, the phenomenon of red brittleness is eliminated by introducing manganese into steel. When the ratio [%Mn]/[%S] = 20 – 22 or more, sulfur is released from the supersaturated solution in the form of manganese sulfides MnS, which have a high melting point.
According to the Sims classification, inclusions of manganese sulfide in cast steel are usually divided into three groups. The appearance of MnS inclusions on the surface of an unetched section is shown in Figure 14.1.
Figure 14.1
– Inclusions of manganese sulfides in cast metal: a – type I manganese sulfides; b – type II manganese sulfides; c – type III manganese sulfides
Type I manganese sulfides are observed in slightly deoxidized steel ([O] > 0.02%). They are globular inclusions of oxysulfides up to 100 microns in size, randomly distributed over the surface of the section.
Type II manganese sulfides are observed when the oxygen content in steel is less than 0.01%. These are homogeneous eutectic sulfides located along grain boundaries in the form of films or chains, as well as sulfides released in the form of flat crystals at the end of the crystallization process.
Manganese III sulfides occur when the oxygen concentration in steel is less than 0.01% and the high content of carbon, silicon, aluminum, etc. These are inclusions with clearly defined edges, corners and planes. They are characterized by apparent homogeneity, but in reality these sulfides contain oxide inclusions.
Currently, aluminum is widely used for metal deoxidation. In this case, manganese sulfides in cast metal are mainly represented by inclusions of types II and III. The peculiarity of these inclusions is their tendency to change shape and size during hot plastic deformation of the metal, as a result of which sulfide inclusions in rolled products have the appearance of long lines elongated in the rolling direction. This is especially typical for steel with a manganese content of 1 - 1.5% or more.
Samples for determining the mechanical properties of the metal can be cut along the rolling direction (longitudinal sample), across (transverse sample) and perpendicular to the rolled surface (vertical sample). Moreover, depending on the orientation of the sample relative to the rolling direction in the section where its destruction occurs, the shape of sulfide inclusions and the area they occupy turn out to be different. The area occupied by sulfide inclusions in the sample destruction section increases when moving from longitudinal to transverse samples and from transverse to vertical samples (Figure 14.2).
Compared to metal, the strength of sulfide inclusions is very low and, to a first rough approximation, they can be considered as cracks that weaken the cross-section of the sample. In this regard, the highest values of the mechanical characteristics of the metal will be established when testing longitudinal samples. When moving to transverse and vertical samples, the mechanical properties of the metal will deteriorate.
Figure 14.2
– Shape of sulfide inclusions in the cross-section of samples cut from rolled stock along the rolling direction, across and perpendicular to the rolled surface
The dependence of the mechanical characteristics of the metal on the relative orientation of sulfide inclusions and the direction of action of the destructive load (or on the orientation of the sample relative to the rolling direction) is called the anisotropy of the mechanical properties of steel. Anisotropy has a particularly strong effect on the plastic properties of the metal - relative elongation, relative contraction, and especially impact strength.
The phenomenon of anisotropy is eliminated by reducing the sulfur concentration in the steel to a low level. As an example, Figure 14.3 shows the effect of sulfur content on the ratio of steel impact strength values obtained by testing transverse, longitudinal and vertical samples cut from seamless pipes. From the data presented it is clear that in order to eliminate the anisotropy of the mechanical characteristics of steel, the sulfur concentration in it should not exceed 0.003 - 0.004%.
Figure 14.3
– Influence of sulfur content on the ratio of steel impact strength on transverse (aq), longitudinal (al) and vertical (ah) samples cut from seamless pipes
Another way to eliminate anisotropy is to modify sulfide inclusions as a result of treatment with alkaline earth and rare earth metals. In these cases, a sufficiently isotropic metal can be obtained with a sulfur content of 0.010 - 0.016%.
In some cases, a higher sulfur content in steel is desirable. An example is free-cut steels, into which sulfur is introduced to produce brittle chips. For example, A12 steel contains, wt%: 0.08 – 0.16 C; 0.6 – 0.9 Mn; 0.15 – 0.35 Si; 0.08 – 0.2 S; no more than 0.15 P.
| 14.1 The role of sulfur in steelmaking processes |
The influence of sulfur on the properties of steels
The sulfur (S) content in high-quality steels does not exceed 0.02-0.03%. In general purpose steels, the permissible sulfur content is higher - 0.03-0.04%. By special treatment of liquid steel, the sulfur content in steel is brought to 0.005%.
Sulfur does not dissolve in iron, so any amount of it forms iron sulfide FeS with iron. This sulfide is part of the eutectic, which is formed at 988 °C.
An increased sulfur content in steels leads to their red brittleness due to low-melting sulfide eutectics that arise along grain boundaries. The phenomenon is red
brittleness occurs at a temperature of 800 °C, that is, at the
red-hot
of steel.
Sulfur has a detrimental effect on the ductility, toughness, weldability and surface quality of steels (especially in steels with low carbon and manganese content).
Sulfur has a very strong tendency to segregate at grain boundaries. This leads to a decrease in the ductility of steels in the hot state. However, 0.08 to 0.33% sulfur is deliberately added to steels for automatic machining. It is known that the presence of sulfur increases the fatigue strength of bearing steels.
The presence of manganese in steel reduces the harmful effects of sulfur. In liquid steel, the formation reaction of manganese sulfide occurs. This sulfide melts at 1620 °C - at temperatures much higher than the temperature of hot working of steels. Manganese sulfides are plastic at temperatures of hot processing of steels (800-1200°C) and therefore are easily deformed.
The influence of carbon, sulfur and phosphorus on steel quality
The determination of carbon, sulfur and phosphorus in steel is of primary importance for metallurgists, foundries and machine builders. This allows you to obtain high-quality products and eliminate irreparable defects. State standards regulate the content of impurities in steel and methods for determining their content.
Carbon in steel
Carbon is a polymorphic non-metallic element that can dissolve in iron in liquid and solid states to form solid solutions - ferrite and austenite. In addition, it creates a chemical compound with iron - cementite (Fe3C), and can be presented in high-carbon steels in the form of graphite.
Depending on the carbon content, steels are classified into:
- low carbon (up to 0.3% C);
- medium carbon (0.3-0.6% C);
- high-carbon (more than 0.6% C).
Carbon content affects the structure of steel, the quantity and ratio of phases, and therefore determines the hardness and ductility of the metal. As the carbon content increases, the impact strength decreases and the cold brittleness threshold increases. An increase in the concentration of C leads to a change in electrical properties: resistance and coercive force increase, magnetic permeability and magnetic induction density decrease.
As carbon increases, casting properties, workability, cutting and weldability deteriorate. Cutting low-carbon steels is also difficult.
Sulfur in steel
Sulfur is a harmful impurity, the main sources of which are pig iron and ore used in steel smelting. It is able to dissolve in liquid iron, and during crystallization it forms FeS. Iron sulfide forms a eutectic with iron with a low melting point, which is located along the grain boundaries. When technologically heated to the temperature of metal pressure treatment, it melts, and when deformed, it becomes the cause of tears and cracks. This phenomenon is called red brittleness, since steel at a temperature of 900-1000℃ becomes bright red.
An increase in sulfur content has a nonlinear effect on the cold brittleness threshold: first it increases, and with an increase in the MnS content it decreases. Sulfur has a negative effect on weldability and corrosion resistance.
Phosphorus in steel
Phosphorus is one of the harmful impurities of steel, the source of which is charge materials, mainly cast iron. It is capable of dissolving in significant quantities in ferrite, which leads to distortion of the crystal lattice. At the same time, there is an increase in tensile strength and yield strength, a decrease in ductility and viscosity. An increase in phosphorus content causes an increase in the cold brittleness threshold and a decrease in the work of crack development.
Phosphorus is highly susceptible to segregation, which leads to a sharp decrease in viscosity in the central part of the ingot. Currently, there is no technology for deep purification of steel from phosphorus.
The influence of aluminum on the properties of steels
Aluminum (Al) is widely used for deoxidation of liquid steel, as well as for grain refinement of steel ingots. The harmful effects of aluminum include the fact that it promotes the graphitization of steels. Although aluminum is often considered an impurity, it is also actively used as an alloying element. Because aluminum forms solid nitrides with nitrogen, it is usually an alloying element in nitrided steels. Aluminum increases the resistance of steels to scaling, and therefore it is added to heat-resistant steels and alloys. In dispersion-hardening stainless steels, aluminum is used as an alloying element that accelerates the dispersion precipitation reaction. Aluminum increases the corrosion resistance of low-carbon steels. Of all the alloying elements, aluminum is the most effective for controlling grain growth when heating steels for hardening.
The influence of nitrogen on the properties of steels
The harmful effect of nitrogen (N) is that the rather large, brittle non-metallic inclusions it forms - nitrides - worsen the properties of steel. A positive property of nitrogen is that it is capable of expanding the austenitic region of the phase diagram of steels. Nitrogen stabilizes the austenitic structure and partially replaces nickel in austenitic steels. Nitride-forming elements vanadium, niobium and titanium are added to low-alloy steels. When controlled by hot working and cooling, they form fine nitrides and carbonitrides, which significantly increase the strength of the steel.
The influence of copper on the properties of steels
Copper (Cu) has a moderate tendency to segregate. The harmful effects of copper include a decrease in the cold brittleness of steel. With an increased copper content, it negatively affects the quality of the steel surface during hot processing. However, with a content of more than 0.20% copper increases its resistance to atmospheric corrosion, as well as the strength properties of alloy and low-alloy steels. Copper in amounts greater than 1% increases the resistance of austenitic stainless steels to sulfuric and hydrochloric acids, as well as their resistance to stress corrosion.
Phosphorus
Iron ores, fuels, and fluxes contain some amount of phosphorus, which during the production of cast iron remains in it to one degree or another and then turns into steel. Phosphorus dissolves well in ferrite and austenite, and at high content it forms phosphide Fe3P (15.62% P). Dissolving in ferrite, phosphorus distorts the crystal lattice and increases the strength and yield limits of steel, greatly reducing ductility and toughness; every 0.01% P increases the cold brittleness threshold by 20...25 0 C. Phosphorus is a harmful impurity in steels.
Like phosphorus, sulfur enters the metal from ores, as well as from furnace gases - a product of fuel combustion (SO2). Sulfur is very limitedly soluble in ferrite, and almost any amount of it forms a sulfur compound with iron - iron sulfide FeS, which is part of the eutectic, which has a melting point of 988 0 C. It is located mainly along the grain boundaries. When steel is heated to rolling or forging temperatures (1000...1200 0 C), the eutectic melts, breaking the bond between grains. During deformation, tears and cracks form in these places. This phenomenon is called red brittleness. The introduction of manganese into steel reduces the harmful effects of sulfur, since when it is introduced into liquid steel, manganese sulfide is formed, which has a melting point
FeS + Mn -> MnS + Fe.
MnS particles are located in the form of separate inclusions and, during deformation, are pulled into lines along the rolling.
Sulfur compounds greatly reduce the mechanical properties of steel under static and cyclic loading, especially toughness, ductility, and endurance limit. Sulfur is a harmful impurity in steels.
Impurities in steels
Steels always contain permanent, harmful and random impurities, since steel is a multicomponent alloy.
Sulfur, phosphorus and all gases are harmful impurities
, and the efforts of metallurgists are always aimed at maximizing the reduction of these elements in steel.
Sulfur
.
The sulfur content in industrial grade steels is usually 0.015...0.050%. Sulfur gives the FeS compound with iron, which forms a low-melting eutectic with iron (melting point 988 ° C), usually located around grains that crystallized before this eutectic. During hot mechanical processing (forging, rolling), the eutectic melts, which causes a loss of connection between the steel grains: the ingot or forging falls apart. This phenomenon is called red brittleness.
Sulfur reduces mechanical properties, especially impact strength and ductility (δ and ψ), as well as fatigue limit. It impairs weldability and corrosion resistance.
Phosphorus.
The phosphorus content in steel is 0.025…0.040%.
Very significantly reduces the viscosity of iron and steel. This harmful effect of phosphorus is especially noticeable at high carbon content in steel and at low temperatures. The phenomenon of increased brittleness of steel at low temperatures is called cold brittleness.
An increase in phosphorus content by every 0.01% increases the cold brittleness threshold by 20...25 °C
.
For some steels, it is possible to increase the sulfur and phosphorus content to improve machinability. This was during the creation of automatic steels, from which fastening materials (nuts, bolts) for non-critical purposes, which are very widely used in mechanical engineering, are produced on high-performance automatic machines. Short, brittle chips and a clean thread surface are the main positive qualities of free-cut steels. Since these steels contain up to 0.15...0.20% sulfur and up to 0.14% phosphorus, such steels can be classified as special.
It is significant that sulfur and phosphorus are strongly eliminated during the crystallization of a steel ingot, resulting in the creation of areas with a sharply increased concentration of these harmful elements compared to their average content in steel.
Gases (nitrogen, oxygen, hydrogen) enter the steel during smelting.
Oxygen, combining with many elements present in steel, forms non-metallic inclusions, so-called oxides (SiO2, Al2O3, etc.). Non-metallic inclusions (oxides, nitrides), being stress concentrators, can significantly reduce the endurance limit and viscosity. Therefore, it is necessary to reduce the oxygen content in steel through good deoxidation and casting in a vacuum, and to carefully control the structure of steel used for the manufacture of critical products.
Hydrogen dissolved in steel is very harmful, as it significantly embrittles the steel. of flakes in rolled billets and forgings
– thin cracks of an oval or round shape, having the appearance of spots in the fracture – silvery flakes.
Permanent impurities
Manganese and silicon are introduced during the steelmaking process for deoxidation; they are technological impurities.
Manganese content
does not exceed 0.8%.
Manganese, present in steel, combines with sulfur more intensively than iron, forming a very refractory compound MnS (melting point 1620 ° C), usually located in the form of small globular inclusions inside steel grains. Consequently, MnS inclusions have a less harmful effect on steel than FeS inclusions .
Silicon content
does not exceed 0.4%. Silicon is a deoxidizer of steel, freeing it from excess oxygen. Silicon dissolves in ferrite and increases the strength of steel, especially the yield strength s0.2 increases. But there is a slight decrease in ductility, which reduces the ability of steel to draw.
The presence of manganese and silicon above the specified limits transfers such steels to the category of special, “alloyed” steels.
Random impurities
– almost any elements accidentally found in steel, for example Cr, Ni, Cu, Mo, Al, Ti, etc., in quantities limited for impurities.
Steels always contain permanent, harmful and random impurities, since steel is a multicomponent alloy.
Sulfur, phosphorus and all gases are harmful impurities
, and the efforts of metallurgists are always aimed at maximizing the reduction of these elements in steel.
Sulfur
.
The sulfur content in industrial grade steels is usually 0.015...0.050%. Sulfur gives the FeS compound with iron, which forms a low-melting eutectic with iron (melting point 988 ° C), usually located around grains that crystallized before this eutectic. During hot mechanical processing (forging, rolling), the eutectic melts, which causes a loss of connection between the steel grains: the ingot or forging falls apart. This phenomenon is called red brittleness.
Sulfur reduces mechanical properties, especially impact strength and ductility (δ and ψ), as well as fatigue limit. It impairs weldability and corrosion resistance.
Phosphorus.
The phosphorus content in steel is 0.025…0.040%.
Very significantly reduces the viscosity of iron and steel. This harmful effect of phosphorus is especially noticeable at high carbon content in steel and at low temperatures. The phenomenon of increased brittleness of steel at low temperatures is called cold brittleness.
An increase in phosphorus content by every 0.01% increases the cold brittleness threshold by 20...25 °C
.
For some steels, it is possible to increase the sulfur and phosphorus content to improve machinability. This was during the creation of automatic steels, from which fastening materials (nuts, bolts) for non-critical purposes, which are very widely used in mechanical engineering, are produced on high-performance automatic machines. Short, brittle chips and a clean thread surface are the main positive qualities of free-cut steels. Since these steels contain up to 0.15...0.20% sulfur and up to 0.14% phosphorus, such steels can be classified as special.
It is significant that sulfur and phosphorus are strongly eliminated during the crystallization of a steel ingot, resulting in the creation of areas with a sharply increased concentration of these harmful elements compared to their average content in steel.
Gases (nitrogen, oxygen, hydrogen) enter the steel during smelting.
Oxygen, combining with many elements present in steel, forms non-metallic inclusions, so-called oxides (SiO2, Al2O3, etc.). Non-metallic inclusions (oxides, nitrides), being stress concentrators, can significantly reduce the endurance limit and viscosity. Therefore, it is necessary to reduce the oxygen content in steel through good deoxidation and casting in a vacuum, and to carefully control the structure of steel used for the manufacture of critical products.
Hydrogen dissolved in steel is very harmful, as it significantly embrittles the steel. of flakes in rolled billets and forgings
– thin cracks of an oval or round shape, having the appearance of spots in the fracture – silvery flakes.
Permanent impurities
Manganese and silicon are introduced during the steelmaking process for deoxidation; they are technological impurities.
Manganese content
does not exceed 0.8%.
Manganese, present in steel, combines with sulfur more intensively than iron, forming a very refractory compound MnS (melting point 1620 ° C), usually located in the form of small globular inclusions inside steel grains. Consequently, MnS inclusions have a less harmful effect on steel than FeS inclusions .
Silicon content
does not exceed 0.4%. Silicon is a deoxidizer of steel, freeing it from excess oxygen. Silicon dissolves in ferrite and increases the strength of steel, especially the yield strength s0.2 increases. But there is a slight decrease in ductility, which reduces the ability of steel to draw.
The presence of manganese and silicon above the specified limits transfers such steels to the category of special, “alloyed” steels.
Random impurities
– almost any elements accidentally found in steel, for example Cr, Ni, Cu, Mo, Al, Ti, etc., in quantities limited for impurities.
Improving steel quality
To remove gases and non-metallic inclusions dissolved in it from liquid steel, vacuum treatment is used. To do this, a ladle with liquid steel is placed in a hermetically sealed chamber, where a vacuum of 267...667 Pa (2...5 mm Hg) is created. The rapidly released gases carry with them non-metallic inclusions and remove them from the metal. Within 10...15 minutes, the amount of dissolved gases decreases by 3...5 times, the number of non-metallic inclusions - by 2...3 times.
To protect the metal from oxidation, steel is cast in an inert atmosphere, for example, argon, under a layer of synthetic slag. To produce steels of particularly high quality, electroslag remelting (ESR), plasma arc remelting, electron beam remelting, and electric arc vacuum remelting are used. The metal is well cleaned (refined) from gases and non-metallic inclusions by treatment with slag and directed crystallization of the liquid melt, creating a deep vacuum.
Dear students! Our website specialists are ready to provide assistance in studying in various subjects: ✔ Solving problems ✔ Completing educational work ✔ Help with exams